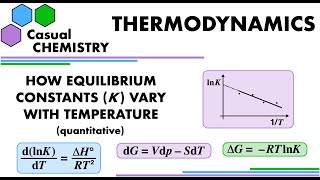
How an Equilibrium Constant varies with Temperature - Thermodynamics - Physical Chemistry
Deriving a quantitative relationship to show how an equilibrium constant varies with temperature and so showing were Le Chatelier's Principle comes from in this context. Along the way, the Gibbs-Helmholtz van't Hoff equations are derived and used. My video for deriving the thermodynamics
From playlist Introductory Thermodynamics

Equilibrium occurs when the overall state of a system is constant. Equilibrium can be static (nothing in the system is changing), or dynamic (little parts of the system are changing, but overall the state isn't changing). In my video, I'll demonstrate systems in both types of equilibrium,
From playlist Physics
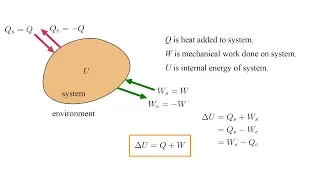
Thermodynamics 3a - Energy and the First Law I
Having developed our ideal-gas model in the previous video, we now use that model to understanding the principle and application of the First Law of Thermodynamics. Note on the definition of a "closed system." I am using the term "closed system" in the sense of the following definition fr
From playlist Thermodynamics

Thermodynamics 3b - Energy and the First Law II
We apply the first law of thermodynamics to understand the Stirling cycle heat engine. Note on the definition of a "closed system." I am using the term "closed system" in the sense of the following definition from Thermal Physics by Charles Kittel: "A closed system is defined as a system
From playlist Thermodynamics

Thermodynamics 4c - Entropy and the Second Law III
We consider in more detail how the fundamental laws of mechanics cannot account for the irreversibility of a system. Yet we find evidence that "special" states are easily transformed into "non-special" states while transforming a non-special state into a special state requires "fine-tuning
From playlist Thermodynamics
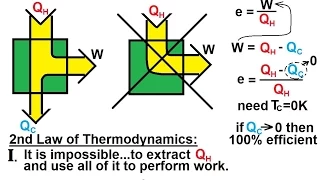
Physics - Thermodynamics: (2 of 14) What is the Second Law of Thermodynamics? (No 100% Efficiency)
Visit http://ilectureonline.com for more math and science lectures! In this video I will explain the 2nd Law of Thermodynamics (no 100% efficiency).
From playlist PHYSICS - THERMODYNAMICS

Universal current fluctuations in non equilibrium systems - Bernard Derrida
Bernard Derrida Ecole Normale Superieure September 20, 2013 Fluctuations of the current of one dimensional non equilibrium diffusive systems are well understood. After a short review of the one dimensional results, the talk will try to show that the statistics of these fluctuations are exa
From playlist Mathematics

Jakob Yngvason - Understanding Entropywithout Probability
Jakob Yngvason (University of Vienna) Understanding Entropy without Probability. In 1856, Rudolf Clausius coined the word entropy as a suitable name for what he had been calling the "transformational content of a body". The new word made it possible to state the second law of thermodynam
From playlist Large-scale limits of interacting particle systems

Thermodynamic System | Open, Closed, Adiabatic, Isolated | Statistical Mechanics
In this video, we will define a thermodynamic system, in particular what kinds of thermodynamic systems there are and how they can interact with their surroundings. References: [1] Ansermet, Brechet, "Principles of Thermodynamics", Cambridge University Press (2019). Follow us on Insta
From playlist Thermodynamics, Statistical Mechanics

On the existence of thermodynamic potentials for quantum systems... by Takahiro Sagawa
PROGRAM CLASSICAL AND QUANTUM TRANSPORT PROCESSES : CURRENT STATE AND FUTURE DIRECTIONS (ONLINE) ORGANIZERS: Alberto Imparato (University of Aarhus, Denmark), Anupam Kundu (ICTS-TIFR, India), Carlos Mejia-Monasterio (Technical University of Madrid, Spain) and Lamberto Rondoni (Polytechn
From playlist Classical and Quantum Transport Processes : Current State and Future Directions (ONLINE)2022

Physics - Thermodynamics 2: Ch 32.1 Def. and Terms (1 of 25) Basic Terms
Visit http://ilectureonline.com for more math and science lectures! In this video I will give and explain the basic terms of thermodynamic heat flow, thermodynamic equilibrium, temperature, system, open system, closed system, and isolated system. Next video in this series can be seen at:
From playlist PHYSICS 32.1 THERMODYNAMICS 2 BASIC TERMS

Courses - G. JONA LASINIO “Macroscopic Fluctuation Theory”
Stationary non-equilibrium states describe steady flows through macroscopic systems. Although they represent the simplest generalization of equilibrium states, they exhibit a variety of new phenomena. Within a statistical mechanics approach, these states have been the subject of several th
From playlist T1-2015 : Disordered systems, random spatial processes and some applications

Surprises in Euler Turbulence: Emergence of Order in 2D Euler Turbulence by Mahendra K Verma
PROGRAM TURBULENCE: PROBLEMS AT THE INTERFACE OF MATHEMATICS AND PHYSICS ORGANIZERS Uriel Frisch (Observatoire de la Côte d'Azur and CNRS, France), Konstantin Khanin (University of Toronto, Canada) and Rahul Pandit (IISc, India) DATE & TIME 16 January 2023 to 27 January 2023 VENUE Ramanuj
From playlist Turbulence: Problems at the Interface of Mathematics and Physics 2023

Self organized tissue architecture: morphogenesis in a dish by Maithreyi Narasimha & Vijaykumar....
Program ICTP - ICTS Winter School on Quantitative Systems Biology ORGANIZERS: Buzz Baum, Guillaume Salbreux, Stefano Di Talia and Vijaykumar Krishnamurthy DATE: 03 December 2019 to 20 December 2019 VENUE: Ramanujan Lecture Hall, ICTS Bangalore The development of an organism from a sin
From playlist Winter School on Quantitative Systems Biology: Quantitative Approaches in Ecosystem Ecology 2020

Thermodynamic limits in cellular information processing by Jeremy Gunawardena
Program Statistical Biological Physics: From Single Molecule to Cell (ONLINE) ORGANIZERS: Debashish Chowdhury (IIT Kanpur), Ambarish Kunwar (IIT Bombay) and Prabal K Maiti (IISc, Bengaluru) DATE: 07 December 2020 to 18 December 2020 VENUE: Online 'Fluctuation-and-noise' are themes
From playlist Statistical Biological Physics: From Single Molecule to Cell (Online)

Gribov Quantization and its Effects on Deconfined Nuclear Matter by Najmul Haque
DISCUSSION MEETING EXTREME NONEQUILIBRIUM QCD (ONLINE) ORGANIZERS: Ayan Mukhopadhyay (IIT Madras) and Sayantan Sharma (IMSc Chennai) DATE & TIME: 05 October 2020 to 09 October 2020 VENUE: Online Understanding quantum gauge theories is one of the remarkable challenges of the millennium
From playlist Extreme Nonequilibrium QCD (Online)

Nonequilibrium Extension of the Third Law of Thermodynamics by Christian Maes
DISCUSSION MEETING : STATISTICAL PHYSICS OF COMPLEX SYSTEMS ORGANIZERS : Sumedha (NISER, India), Abhishek Dhar (ICTS-TIFR, India), Satya Majumdar (University of Paris-Saclay, France), R Rajesh (IMSc, India), Sanjib Sabhapandit (RRI, India) and Tridib Sadhu (TIFR, India) DATE : 19 December
From playlist Statistical Physics of Complex Systems - 2022

Scaling down the laws of thermodynamics by Christopher Jarzynski
PROGRAM : FLUCTUATIONS IN NONEQUILIBRIUM SYSTEMS: THEORY AND APPLICATIONS ORGANIZERS : Urna Basu and Anupam Kundu DATE : 09 March 2020 to 19 March 2020 VENUE : Madhava Lecture Hall, ICTS, Bangalore THIS PROGRAM HAS BEEN MODIFIED ONLY FOR LOCAL (BANGALORE) PARTICIPANTS DUE TO COVID-19 RI
From playlist Fluctuations in Nonequilibrium Systems: Theory and Applications

Thermodynamic Uncertainty Relations by Supriya Krishnamurthy
DISCUSSION MEETING : STATISTICAL PHYSICS OF COMPLEX SYSTEMS ORGANIZERS : Sumedha (NISER, India), Abhishek Dhar (ICTS-TIFR, India), Satya Majumdar (University of Paris-Saclay, France), R Rajesh (IMSc, India), Sanjib Sabhapandit (RRI, India) and Tridib Sadhu (TIFR, India) DATE : 19 December
From playlist Statistical Physics of Complex Systems - 2022

Kirone Mallick - Integrability and non-equilibrium statistical physics
During the last twenty years, a large number of exact solutions have been derived for some non-equilibrium interacting systems, such as the exclusion process, leading us to a better understanding of non-equilibrium behaviour. Integrability has played an important role in these developments
From playlist 6e Séminaire Itzykson : "Physique statistique hors équilibre"