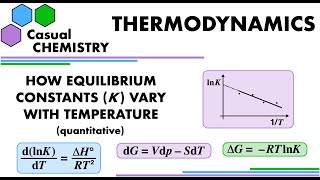
How an Equilibrium Constant varies with Temperature - Thermodynamics - Physical Chemistry
Deriving a quantitative relationship to show how an equilibrium constant varies with temperature and so showing were Le Chatelier's Principle comes from in this context. Along the way, the Gibbs-Helmholtz van't Hoff equations are derived and used. My video for deriving the thermodynamics
From playlist Introductory Thermodynamics

Thermodynamic System | Open, Closed, Adiabatic, Isolated | Statistical Mechanics
In this video, we will define a thermodynamic system, in particular what kinds of thermodynamic systems there are and how they can interact with their surroundings. References: [1] Ansermet, Brechet, "Principles of Thermodynamics", Cambridge University Press (2019). Follow us on Insta
From playlist Thermodynamics, Statistical Mechanics

Equilibrium occurs when the overall state of a system is constant. Equilibrium can be static (nothing in the system is changing), or dynamic (little parts of the system are changing, but overall the state isn't changing). In my video, I'll demonstrate systems in both types of equilibrium,
From playlist Physics

Physics - Thermodynamics 2: Ch 32.1 Def. and Terms (7 of 23) Visiual Rep. of Equilibrium State
Visit http://ilectureonline.com for more math and science lectures! In this video I will give and explain a visual representation of equilibrium state and equation of state. Next video in this series can be seen at: https://youtu.be/UHjnWqmt_OA
From playlist PHYSICS 32.1 THERMODYNAMICS 2 BASIC TERMS

Physics - Thermodynamics 2: Ch 32.1 Def. and Terms (1 of 25) Basic Terms
Visit http://ilectureonline.com for more math and science lectures! In this video I will give and explain the basic terms of thermodynamic heat flow, thermodynamic equilibrium, temperature, system, open system, closed system, and isolated system. Next video in this series can be seen at:
From playlist PHYSICS 32.1 THERMODYNAMICS 2 BASIC TERMS
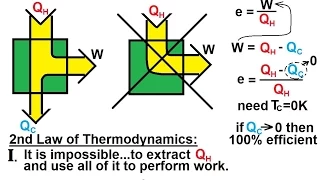
Physics - Thermodynamics: (2 of 14) What is the Second Law of Thermodynamics? (No 100% Efficiency)
Visit http://ilectureonline.com for more math and science lectures! In this video I will explain the 2nd Law of Thermodynamics (no 100% efficiency).
From playlist PHYSICS - THERMODYNAMICS
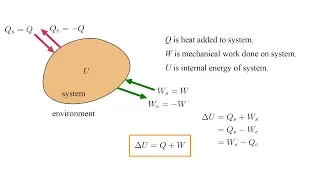
Thermodynamics 3a - Energy and the First Law I
Having developed our ideal-gas model in the previous video, we now use that model to understanding the principle and application of the First Law of Thermodynamics. Note on the definition of a "closed system." I am using the term "closed system" in the sense of the following definition fr
From playlist Thermodynamics

Teach Astronomy - Thermal Equilibrium
http://www.teachastronomy.com/ Thermal equilibrium is an important physical principle. When two substances have unequal temperatures, thermal equilibrium means that they will tend to evolve to a situation of equal temperature. At a microscopic level we know that temperature denotes the in
From playlist 04. Chemistry and Physics

Thermodynamics 4a - Entropy and the Second Law I
The Second Law of Thermodynamics is one of the most important laws in all of physics. But it is also one of the more difficult to understand. Central to it are the concepts of reversibility and entropy. Note on the definition of a "closed system." I am using the term "closed system" in th
From playlist Thermodynamics

Jakob Yngvason - Understanding Entropywithout Probability
Jakob Yngvason (University of Vienna) Understanding Entropy without Probability. In 1856, Rudolf Clausius coined the word entropy as a suitable name for what he had been calling the "transformational content of a body". The new word made it possible to state the second law of thermodynam
From playlist Large-scale limits of interacting particle systems

On the existence of thermodynamic potentials for quantum systems... by Takahiro Sagawa
PROGRAM CLASSICAL AND QUANTUM TRANSPORT PROCESSES : CURRENT STATE AND FUTURE DIRECTIONS (ONLINE) ORGANIZERS: Alberto Imparato (University of Aarhus, Denmark), Anupam Kundu (ICTS-TIFR, India), Carlos Mejia-Monasterio (Technical University of Madrid, Spain) and Lamberto Rondoni (Polytechn
From playlist Classical and Quantum Transport Processes : Current State and Future Directions (ONLINE)2022

Scaling down the laws of thermodynamics by Christopher Jarzynski
PROGRAM : FLUCTUATIONS IN NONEQUILIBRIUM SYSTEMS: THEORY AND APPLICATIONS ORGANIZERS : Urna Basu and Anupam Kundu DATE : 09 March 2020 to 19 March 2020 VENUE : Madhava Lecture Hall, ICTS, Bangalore THIS PROGRAM HAS BEEN MODIFIED ONLY FOR LOCAL (BANGALORE) PARTICIPANTS DUE TO COVID-19 RI
From playlist Fluctuations in Nonequilibrium Systems: Theory and Applications

Self organized tissue architecture: morphogenesis in a dish by Maithreyi Narasimha & Vijaykumar....
Program ICTP - ICTS Winter School on Quantitative Systems Biology ORGANIZERS: Buzz Baum, Guillaume Salbreux, Stefano Di Talia and Vijaykumar Krishnamurthy DATE: 03 December 2019 to 20 December 2019 VENUE: Ramanujan Lecture Hall, ICTS Bangalore The development of an organism from a sin
From playlist Winter School on Quantitative Systems Biology: Quantitative Approaches in Ecosystem Ecology 2020

Engineering MAE 91. Intro to Thermodynamics. Lecture 02.
UCI MAE 91: Introduction to Thermodynamics (Spring 2013). Lec 02. Intro to Thermodynamics -- Thermodynamic Properties -- View the complete course: http://ocw.uci.edu/courses/mae_91_introduction_to_thermal_dynamics.html Instructor: Roger Rangel, Ph.D. License: Creative Commons CC-BY-SA Ter
From playlist Engineering MAE 91. Intro to Thermodynamics

Thermodynamics and Chemical Dynamics 131C. Lecture 27. The Final Exam
UCI Chem 131C Thermodynamics and Chemical Dynamics (Spring 2012) Lec 27. Thermodynamics and Chemical Dynamics -- The Final Exam -- View the complete course: http://ocw.uci.edu/courses/chem_131c_thermodynamics_and_chemical_dynamics.html Instructor: Reginald Penner, Ph.D. License: Creative
From playlist Chemistry 131C: Thermodynamics and Chemical Dynamics

Zeroth law of thermodynamics | Chemical Processes | MCAT | Khan Academy
Visit us (http://www.khanacademy.org/science/healthcare-and-medicine) for health and medicine content or (http://www.khanacademy.org/test-prep/mcat) for MCAT related content. These videos do not provide medical advice and are for informational purposes only. The videos are not intended to
From playlist Chemical processes | MCAT | Khan Academy

Lec 1 | MIT 5.60 Thermodynamics & Kinetics, Spring 2008
Lecture 1: State of a system, 0th law, equation of state. View the complete course at: http://ocw.mit.edu/5-60S08 License: Creative Commons BY-NC-SA More information at http://ocw.mit.edu/terms More courses at http://ocw.mit.edu
From playlist MIT 5.60 Thermodynamics & Kinetics, Spring 2008

Courses - G. JONA LASINIO “Macroscopic Fluctuation Theory”
Stationary non-equilibrium states describe steady flows through macroscopic systems. Although they represent the simplest generalization of equilibrium states, they exhibit a variety of new phenomena. Within a statistical mechanics approach, these states have been the subject of several th
From playlist T1-2015 : Disordered systems, random spatial processes and some applications

Physics - Thermodynamics: Radiation: Heat Transfer (8 of 11) Earth's Equilibrium Temperature
Visit http://ilectureonline.com for more math and science lectures! In this video I will show you how to calculate the Earth's equilibrium temperature.
From playlist PHYSICS - THERMODYNAMICS