
Thermodynamic System | Open, Closed, Adiabatic, Isolated | Statistical Mechanics
In this video, we will define a thermodynamic system, in particular what kinds of thermodynamic systems there are and how they can interact with their surroundings. References: [1] Ansermet, Brechet, "Principles of Thermodynamics", Cambridge University Press (2019). Follow us on Insta
From playlist Thermodynamics, Statistical Mechanics

How a bimetallic thermostat works!
Most things get bigger when they heat up and smaller when they cool down. Mechanical thermostats use this idea (which is called thermal expansion) to switch an electric circuit on and and off.
From playlist THERMODYNAMICS

AWESOME Physics demonstrations. Thermal expansion of gases and fluids!
The air in the container and the balloon extends during heating. The balloon also gets bigger. When the air is cooled down, its volume decreases and so decreases also the volume of the balloon. The same happens to the liquid on a bottle. This is physics experiment for science project.
From playlist THERMODYNAMICS

How thermoelectric generator works!
I show you how to build a Thermoelectric Generator. This generator converts heat directly into electricity.
From playlist THERMODYNAMICS

Thermodynamics: What do HEAT and WORK really mean? | Basics of Thermodynamics
"Work" and "heat" are commonly used words in everyday life. But they mean very specific things in the physics field of thermodynamics. Hey everyone, in this (rather short) video I wanted to discuss the exact definitions of the words "work" and "heat" in a thermodynamic context. These word
From playlist Thermodynamics by Parth G

Physics - Thermodynamics: (1 of 14) Efficiency Of Heat Engines: Basics
Visit http://ilectureonline.com for more math and science lectures! In this video I will explain the efficiency of the heat engine.
From playlist PHYSICS - THERMODYNAMICS
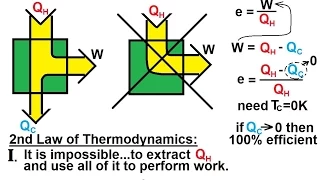
Physics - Thermodynamics: (2 of 14) What is the Second Law of Thermodynamics? (No 100% Efficiency)
Visit http://ilectureonline.com for more math and science lectures! In this video I will explain the 2nd Law of Thermodynamics (no 100% efficiency).
From playlist PHYSICS - THERMODYNAMICS

In the first mini-lecture, we begin by asking ‘what is thermodynamics?’ We understand it as the measurement of the relationship between heat, work, energy and temperature. When considering chemical reactions, we look at the 1st law of thermodynamics, and how this relates to the two differe
From playlist Chemistry

Thermodynamics 3b - Energy and the First Law II
We apply the first law of thermodynamics to understand the Stirling cycle heat engine. Note on the definition of a "closed system." I am using the term "closed system" in the sense of the following definition from Thermal Physics by Charles Kittel: "A closed system is defined as a system
From playlist Thermodynamics

Matteo Tanzi - Self-consistent transfer operators for high-dimensional expanding coupled maps
Recorded 29 August 2022. Matteo Tanzi of New York University presents "Self-consistent transfer operators for high-dimensional expanding coupled maps" at IPAM's Reconstructing Network Dynamics from Data: Applications to Neuroscience and Beyond. Abstract: Recently, much progress has been ma
From playlist 2022 Reconstructing Network Dynamics from Data: Applications to Neuroscience and Beyond

On the existence of thermodynamic potentials for quantum systems... by Takahiro Sagawa
PROGRAM CLASSICAL AND QUANTUM TRANSPORT PROCESSES : CURRENT STATE AND FUTURE DIRECTIONS (ONLINE) ORGANIZERS: Alberto Imparato (University of Aarhus, Denmark), Anupam Kundu (ICTS-TIFR, India), Carlos Mejia-Monasterio (Technical University of Madrid, Spain) and Lamberto Rondoni (Polytechn
From playlist Classical and Quantum Transport Processes : Current State and Future Directions (ONLINE)2022

Mod-01 Lec-13 Thermodynamics of Turbines
Jet Aircraft Propulsion by Prof. Bhaskar Roy and Prof. A. M. Pradeep, Department of Aerospace Engineering, IIT Bombay. For more details on NPTEL visit http://nptel.iitm.ac.in
From playlist IIT Bombay: Aerospace - Jet Aircraft Propulsion (CosmoLearning Aerospace Engineering)

Engineering MAE 91. Intro to Thermodynamics. Lecture 11.
UCI MAE 91: Introduction to Thermodynamics (Spring 2013). Lec 08. Intro to Thermodynamics -- First Law of Thermodynamics for Open Systems -- View the complete course: http://ocw.uci.edu/courses/mae_91_introduction_to_thermal_dynamics.html Instructor: Roger Rangel, Ph.D. License: Creative
From playlist Engineering MAE 91. Intro to Thermodynamics

Structure-preserving numerical approximations to thermodynamically consistent dissipative models
Qi Wang University of South Carolina, USA Beijing Computational Science Research Center, China
From playlist 2018 Modeling and Simulation of Interface Dynamics in Fluids/Solids and Their Applications

Serena Cenatiempo - Universal behavior of Bose-Einstein condensates in the Gross-Pitaevskii regime
Serena Cenatiempo (Gran Sasso Science Institute) Universal behavior of Bose-Einstein condensates in the Gross-Pitaevskii regime. We consider systems of N interacting bosons in the Gross-Pitaevskii limit, where both the range and the scattering length of the potential are of the order 1/N
From playlist Large-scale limits of interacting particle systems

The equilibrium landscape of the Heisenberg spin chain by Enej Ilievski
PROGRAM THERMALIZATION, MANY BODY LOCALIZATION AND HYDRODYNAMICS ORGANIZERS: Dmitry Abanin, Abhishek Dhar, François Huveneers, Takahiro Sagawa, Keiji Saito, Herbert Spohn and Hal Tasaki DATE : 11 November 2019 to 29 November 2019 VENUE: Ramanujan Lecture Hall, ICTS Bangalore How do is
From playlist Thermalization, Many Body Localization And Hydrodynamics 2019

Gary Froyland: Spectral perturbation formulae for quenched random dynamics with applications to...
CONFERENCE Recording during the thematic meeting : "Probabilistic Techniques for Random and Time-Dependent Dynamical Systems" the October 6, 2022 at the Centre International de Rencontres Mathématiques (Marseille, France) Filmmaker: Guillaume Hennenfent Find this video and other talks g
From playlist Dynamical Systems and Ordinary Differential Equations

Jakob Yngvason - Understanding Entropywithout Probability
Jakob Yngvason (University of Vienna) Understanding Entropy without Probability. In 1856, Rudolf Clausius coined the word entropy as a suitable name for what he had been calling the "transformational content of a body". The new word made it possible to state the second law of thermodynam
From playlist Large-scale limits of interacting particle systems

Dynamical phase transitions, eigenstate thermalization, and Schrodinger cats... - David A. Huse
David A. Huse Princeton University October 15, 2013 An isolated quantum many-body system may be a reservoir that thermalizes its constituents. I will explore an example of the interplay of this thermalization and spontaneous symmetry-breaking, in the ferromagnetic phase of an infinite-rang
From playlist Mathematics

Physics - Thermodynamics 2: Ch 32.1 Def. and Terms (1 of 25) Basic Terms
Visit http://ilectureonline.com for more math and science lectures! In this video I will give and explain the basic terms of thermodynamic heat flow, thermodynamic equilibrium, temperature, system, open system, closed system, and isolated system. Next video in this series can be seen at:
From playlist PHYSICS 32.1 THERMODYNAMICS 2 BASIC TERMS