Degree of ionization
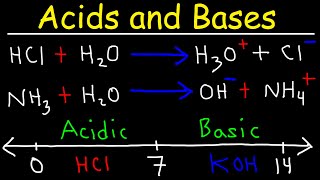
The degree of ionization (also known as ionization yield in the literature) refers to the proportion of neutral particles, such as those in a gas or aqueous solution, that are ionized. For electrolytes, it could be understood as a capacity of acid/base to ionize itself. A low degree of ionization is sometimes called partially ionized (also weakly ionized), and a high degree of ionization as fully ionized. However, fully ionized can also mean that an ion has no electrons left. Ionization refers to the process whereby an atom or molecule loses one or several electrons from its atomic orbital, or conversely gains an additional one, from an incoming free electron (electron attachment). In both cases, the atom or molecule ceases to be a neutral particle and becomes a charge carrier. If the species has lost one or several electrons, it becomes positively charged and is called a positive ion, or cation. On the contrary, if the species has gained one or several additional electrons, it becomes negatively charged and is called a negative ion, or anion. Individual free electrons and ions in a plasma have very short lives typically inferior to the microsecond, as ionization and recombination, excitation and relaxation are collective continuous processes. (Wikipedia).