Thermodynamics 3c - Energy and the First Law III
Here we complete our discussion of the First Law and thermodynamic cycles. Note on the definition of a "closed system." I am using the term "closed system" in the sense of the following definition from Thermal Physics by Charles Kittel: "A closed system is defined as a system with constan
From playlist Thermodynamics

Dynamical phase transitions, eigenstate thermalization, and Schrodinger cats... - David A. Huse
David A. Huse Princeton University October 15, 2013 An isolated quantum many-body system may be a reservoir that thermalizes its constituents. I will explore an example of the interplay of this thermalization and spontaneous symmetry-breaking, in the ferromagnetic phase of an infinite-rang
From playlist Mathematics
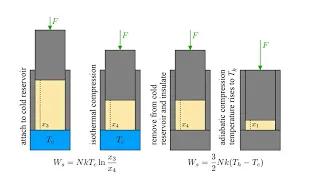
Thermodynamics 4b - Entropy and the Second Law II
We compare the reversibility of the Carnot cycle to the irreversibility of the Stirling cycle and find that they may be accounted for by the constancy or increase of transferred heat divided by temperature. We then consider how conservation laws, including the fundamental laws of mechanics
From playlist Thermodynamics

How Refrigerators Work? (Cooling Systems Explained)
This video shows how cooling systems works, like refrigerators and air conditioners. By Analyzing the thermodynamic processes that happens inside those systems, for one of the most widely used cycles. It also shows what happens inside the main parts of the cooling system(condenser, compre
From playlist Physics

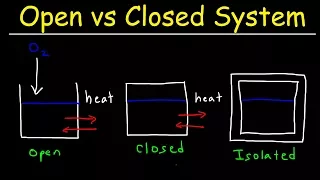
Thermodynamic System | Open, Closed, Adiabatic, Isolated | Statistical Mechanics
In this video, we will define a thermodynamic system, in particular what kinds of thermodynamic systems there are and how they can interact with their surroundings. References: [1] Ansermet, Brechet, "Principles of Thermodynamics", Cambridge University Press (2019). Follow us on Insta
From playlist Thermodynamics, Statistical Mechanics

Chemistry - Thermochemistry (1 of 37) Definition
Visit http://ilectureonline.com for more math and science lectures! In this video I will introduce of the definition of thermochemistry.
From playlist MOST POPULAR VIDEOS

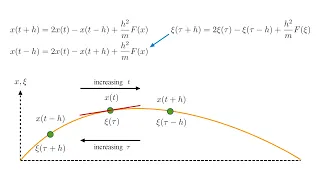
Thermodynamics 3b - Energy and the First Law II
We apply the first law of thermodynamics to understand the Stirling cycle heat engine. Note on the definition of a "closed system." I am using the term "closed system" in the sense of the following definition from Thermal Physics by Charles Kittel: "A closed system is defined as a system
From playlist Thermodynamics
Open System, Closed System and Isolated System - Thermodynamics & Physics
This physics video tutorial provides a basic introduction into open systems, closed systems, and isolated systems. New Physics Video Playlist: https://www.youtube.com/playlist?list=PL0o_zxa4K1BU6wPPLDsoTj1_wEf0LSNeR Access to Premium Videos: https://www.patreon.com/MathScienceTutor ht
From playlist New Physics Video Playlist

Physics - Thermodynamics 2: Ch 32.1 Def. and Terms (1 of 25) Basic Terms
Visit http://ilectureonline.com for more math and science lectures! In this video I will give and explain the basic terms of thermodynamic heat flow, thermodynamic equilibrium, temperature, system, open system, closed system, and isolated system. Next video in this series can be seen at:
From playlist PHYSICS 32.1 THERMODYNAMICS 2 BASIC TERMS
MIT RES.TLL-004 Concept Vignettes View the complete course: http://ocw.mit.edu/RES-TLL-004F13 Instructor: John Lienhard In this video, four scenarios are presented wherein the heat transfer between a pan and its handle, and between the handle and the room are constrained in a variety of w
From playlist MIT STEM Concept Videos

Thermalization in closed quantum many-body systems I: Basic notions by Stefan Kehrein
Open Quantum Systems DATE: 17 July 2017 to 04 August 2017 VENUE: Ramanujan Lecture Hall, ICTS Bangalore There have been major recent breakthroughs, both experimental and theoretical, in the field of Open Quantum Systems. The aim of this program is to bring together leaders in the Open Q
From playlist Open Quantum Systems

Ergodicity breaking in quantum many-body systems by Sthitadhi Roy
COLLOQUIUM ERGODICITY BREAKING IN QUANTUM MANY-BODY SYSTEMS SPEAKER: Sthitadhi Roy (University of Oxford, UK) DATE: Mon, 31 May 2021, 15:30 to 17:00 VENUE: Online Colloquium ABSTRACT Ergodicity is a key ingredient in how thermodynamics emerges from microscopic descriptions of compl
From playlist ICTS Colloquia

15.2 The Zeroth Law of Thermodynamics
This video covers Section 15.2 of Cutnell & Johnson Physics 10e, by David Young and Shane Stadler, published by John Wiley and Sons. The lecture is part of the course General Physics - Life Sciences I and II, taught by Dr. Boyd F. Edwards at Utah State University. This video was produced
From playlist Lecture 15A. Thermodynamics

Peter R Saulson - Vibration isolation (Passive systems)
PROGRAM: ICTS Winter School on Experimental Gravitational-Wave Physics DATES: Monday 23 Dec, 2013 - Saturday 28 Dec, 2013 VENUE: Raja Ramanna Centre for Advanced Technology, Indore PROGRAM LINK: http://www.icts.res.in/program/GWS2013 A worldwide network of detectors are currently involved
From playlist ICTS Winter School on Experimental Gravitational-Wave Physics

Lec 31b - Phys 237: Gravitational Waves with Kip Thorne
Watch the rest of the lectures on http://www.cosmolearning.com/courses/overview-of-gravitational-wave-science-400/ Redistributed with permission. This video is taken from a 2002 Caltech on-line course on "Gravitational Waves", organized and designed by Kip S. Thorne, Mihai Bondarescu and
From playlist Caltech: Gravitational Waves with Kip Thorne - CosmoLearning.com Physics

EEVblog #744 - SMD Thermal Heatsink Design - µSupply Part 15
Dave explains how to attach an SMD power transistor or regulator to a case to use as a heat sink in this design tutorial. And in the process talks about thermal design, the electrical/thermal analogy, and thermal vias. This is Part 15 of the µSupply Power supply design series. Other video
From playlist Thermal Design

Surprises in Euler Turbulence: Emergence of Order in 2D Euler Turbulence by Mahendra K Verma
PROGRAM TURBULENCE: PROBLEMS AT THE INTERFACE OF MATHEMATICS AND PHYSICS ORGANIZERS Uriel Frisch (Observatoire de la Côte d'Azur and CNRS, France), Konstantin Khanin (University of Toronto, Canada) and Rahul Pandit (IISc, India) DATE & TIME 16 January 2023 to 27 January 2023 VENUE Ramanuj
From playlist Turbulence: Problems at the Interface of Mathematics and Physics 2023

Thermodynamics 4a - Entropy and the Second Law I
The Second Law of Thermodynamics is one of the most important laws in all of physics. But it is also one of the more difficult to understand. Central to it are the concepts of reversibility and entropy. Note on the definition of a "closed system." I am using the term "closed system" in th
From playlist Thermodynamics

Winter Theory School 2022: Anushya Chandran Talk Two
Many-body Locatlization
From playlist Winter Theory 2022
