
Physics - Ch 66.5 Quantum Mechanics: The Hydrogen Atom (42 of 78) What is the Rydberg Constant?
Visit http://ilectureonline.com for more math and science lectures! In this video I will find the Rydberg constant and its relationship to the reduced mass. I will use the equation used to find the wavelength of the photon created by the electron jumps in the hydrogen atom. The wave equat
From playlist THE "WHAT IS" PLAYLIST

#MegaFavNumbers Rydberg's Constant
The Rydberg constant is used to calculate the wavelengths of light emitted by hydrogen. #MegaFavNumbers Note: If you are on a small screen the text may be a bit blurry.
From playlist MegaFavNumbers

Watch more videos on http://www.brightstorm.com/science/physics SUBSCRIBE FOR All OUR VIDEOS! https://www.youtube.com/subscription_center?add_user=brightstorm2 VISIT BRIGHTSTORM.com FOR TONS OF VIDEO TUTORIALS AND OTHER FEATURES! http://www.brightstorm.com/ LET'S CONNECT! Facebook ► ht
From playlist Physics

Physics - Thermodynamics 2: Ch 32.1 Def. and Terms (9 of 23) What is the Gas Constant?
Visit http://ilectureonline.com for more math and science lectures! In this video I will give and explain what is the gas constant and how it was determined. Next video in this series can be seen at: https://youtu.be/8N8TN0L5xiQ
From playlist PHYSICS 32.1 THERMODYNAMICS 2 BASIC TERMS

Emission spectrum of hydrogen | Chemistry | Khan Academy
Using Balmer-Rydberg equation to solve for photon energy for n=3 to 2 transition. Solving for wavelength of a line in UV region of hydrogen emission spectrum. Created by Jay. Watch the next lesson: https://www.khanacademy.org/science/chemistry/electronic-structure-of-atoms/orbitals-and-e
From playlist Quantum Physics | AP Physics 2 | Khan Academy

Emission spectrum of hydrogen | Physical Processes | MCAT | Khan Academy
Visit us (http://www.khanacademy.org/science/healthcare-and-medicine) for health and medicine content or (http://www.khanacademy.org/test-prep/mcat) for MCAT related content. These videos do not provide medical advice and are for informational purposes only. The videos are not intended to
From playlist Physical processes | MCAT | Khan Academy

Teach Astronomy - Measuring the Hubble Constant
http://www.teachastronomy.com/ The Hubble constant sets the current expansion rate of the universe and gives an indication of its size and age. The best currently measured value of the Hubble constant comes from a heroic project done with the Hubble Space Telescope over a number of years.
From playlist 20. Galaxy Interaction and Motion

General Chemistry 1A. Lecture 04. Wavelengths.
UCI Chem 1A General Chemistry (Winter 2013) Lec 04. General Chemistry Wavelengths View the complete course: http://ocw.uci.edu/courses/chem_1a_general_chemistry.html Instructor: Amanda Brindley, Ph.D. License: Creative Commons BY-NC-SA Terms of Use: http://ocw.uci.edu/info. More courses
From playlist Chem 1A: General Chemistry

Determining when a function is increasing decreasing or constant
👉 Learn how to determine increasing/decreasing intervals. There are many ways in which we can determine whether a function is increasing or decreasing but we will focus on determining increasing/decreasing from the graph of the function. A function is increasing when the graph of the funct
From playlist When is the Function Increasing Decreasing or Neither

5. Hydrogen atom energy levels
MIT 5.111 Principles of Chemical Science, Fall 2008 View the complete course: http://ocw.mit.edu/5-111F08 Instructor: Catherine Drennan, Elizabeth Vogel Taylor License: Creative Commons BY-NC-SA More information at http://ocw.mit.edu/terms More courses at http://ocw.mit.edu
From playlist MIT 5.111 Principles of Chemical Science, Fall 2008

Relative Maxima and Minima(HD)
Watch more videos on http://www.brightstorm.com/math/calculus SUBSCRIBE FOR All OUR VIDEOS! https://www.youtube.com/subscription_center?add_user=brightstorm2 VISIT BRIGHTSTORM.com FOR TONS OF VIDEO TUTORIALS AND OTHER FEATURES! http://www.brightstorm.com/ LET'S CONNECT! Facebook ► https
From playlist Calculus

Absorption and emission | Electronic structure of atoms | Chemistry | Khan Academy
Calculating electron energy for levels n=1 to 3. Drawing a shell model diagram and an energy diagram for hydrogen, and then using the diagrams to calculate the energy required to excite an electron between different energy levels. Created by Jay. Watch the next lesson: https://www.khanac
From playlist Quantum Physics | AP Physics 2 | Khan Academy

Intervals of increasing and decreasing function from a graph
👉 Learn how to determine increasing/decreasing intervals. There are many ways in which we can determine whether a function is increasing or decreasing but we will focus on determining increasing/decreasing from the graph of the function. A function is increasing when the graph of the funct
From playlist When is the Function Increasing Decreasing or Neither

The Best Scientific Measurements and How We Use Them
Some specific measurements have helped scientists make amazing discoveries. Over the years, scientists have made measurements proving that we understand ridiculously well how the universe works. So what are they? Join Hank Green for a fascinating new episode of SciShow! SciShow has a spi
From playlist Uploads

Ionisation Energy for the First 10 Elements (Physical Chemistry, Physics)
A consideration of how much an outer electron feels the effect of a nuclear charge after shielding effects of other electrons. quantum mechanical correction for exchange energy consideration due to exact electron configurations is then explored. #chemistry #pchem #atom #quantum #physicalc
From playlist Random Physical Chemistry
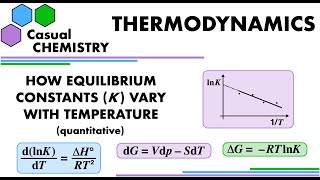
How an Equilibrium Constant varies with Temperature - Thermodynamics - Physical Chemistry
Deriving a quantitative relationship to show how an equilibrium constant varies with temperature and so showing were Le Chatelier's Principle comes from in this context. Along the way, the Gibbs-Helmholtz van't Hoff equations are derived and used. My video for deriving the thermodynamics
From playlist Introductory Thermodynamics

Physics - Ch 66.5 Quantum Mechanics: The Hydrogen Atom (43 of 78) The Reduced Mass Correction
Visit http://ilectureonline.com for more math and science lectures! In this video I will find the wavelength=? of H-alpha lines on the reduced mass on the wavelengths of the photons emitted when there is an energy jump from the 3rd level down to the 2nd level in the hydrogen atom. Next v
From playlist PHYSICS 66.5 QUANTUM MECHANICS: THE HYDROGEN ATOM

MIT 8.421 Atomic and Optical Physics I, Spring 2014 View the complete course: http://ocw.mit.edu/8-421S14 Instructor: Wolfgang Ketterle In this lecture, the professor talked about the atomic units, atomic structure, helium atom and energy levels of helium. License: Creative Commons BY-NC
From playlist MIT 8.421 Atomic and Optical Physics I, Spring 2014

Physics - Ch 66.5 Quantum Mechanics: The Hydrogen Atom (10 of 78) Electron Transition in Hydrogen
Visit http://ilectureonline.com for more math and science lectures! In this video I will calculate the Hydberg constant and from that find the inverse of the wavelength of the absorbed or emitted energy(because base on the Uncertain Principle electrons can “jump” from one orbit to another
From playlist PHYSICS 66.5 QUANTUM MECHANICS: THE HYDROGEN ATOM
The Normal Distribution (1 of 3: Introductory definition)
More resources available at www.misterwootube.com
From playlist The Normal Distribution