
Bohr Model (5 of 7) Bohr Radius Derivation
This video shows how to derive the Bohr radius of a hydrogen atom and the radii for the additional excited states. The Bohr radius is a physical constant, approximately equal to the most probable distance between the nucleus and the electron in a hydrogen atom in its ground state. It is n
From playlist Quantum Mechanics

Physics - Modern Physics (15 of 26) The Bohr Atom: Radius of the Atom
Visit http://ilectureonline.com for more math and science lectures! In this video I will show you how find the radius of the Bohr Atom (hydrogen atom).
From playlist MOST POPULAR VIDEOS

Physics - Modern Physics (17 of 26) The Bohr Atom: Velocity and Radius of the Electron
Visit http://ilectureonline.com for more math and science lectures! In this video I will show you how find the velocity and radius of the electron of the Bohr Atom
From playlist MODERN PHYSICS 2: ATOMIC AND NUCLEAR PHYSICS, PARTICLE PHYSICS

Physics - Ch 66.5 Quantum Mechanics: The Hydrogen Atom (6 of 78) How was the Bohr Radius Determined2
Visit http://ilectureonline.com for more math and science lectures! In this video I will calculate and explain how the Bohr radius was determined. The small electron with a high velocity v traveling like a wave around the nucleus of an atom will have the path length equaling an integer of
From playlist PHYSICS 66.5 QUANTUM MECHANICS: THE HYDROGEN ATOM

How Quantum Physics Predicts the Size of an Atom (Bohr Radius) by Parth G
Atoms are tiny particles that make up everything. But exactly how big (or rather how small) are they? In this video, we'll start by defining the "size" of an atom as the distance between the center of the nucleus, and the furthest (outermost) electron. This can also be known as the radius
From playlist Quantum Physics by Parth G
The Atom A5 The Bohr Model of the Hydrogen Atom
The Bohr model of the atom.
From playlist Physics - The Atom

Physics - Modern Physics (14 of 26) The Bohr Atom (Part I)
Visit http://ilectureonline.com for more math and science lectures! In this video I will explain the Bohr Atom and find the equation for its energy.
From playlist MOST POPULAR VIDEOS

The Atom A4 The Bohr Model of the Hydrogen Atom
The Bohr model of the atom.
From playlist Physics - The Atom

The Atom A3 The Bohr Model of the Hydrogen Atom
The Bohr model of the atom.
From playlist Physics - The Atom

Bohr model radii | Electronic structure of atoms | Chemistry | Khan Academy
Using equation for Bohr model radii to draw shell model for n=1 to 3, and calculating the velocity of a ground state electron. Created by Jay. Watch the next lesson: https://www.khanacademy.org/science/chemistry/electronic-structure-of-atoms/bohr-model-hydrogen/v/bohr-model-energy-le
From playlist Quantum Physics | AP Physics 2 | Khan Academy
Chemistry - Electron Structures in Atoms (15 of 40) The Bohr Atom & Particle Wave Duality
Visit http://ilectureonline.com for more math and science lectures! In this video I will explain the Bohr atom and particle duality.
From playlist CHEMISTRY 11 ELECTRON ORBITALS AND ATOMIC STRUCTURE

What is the Bohr model of the atom?
This video looks at the pioneering work of Niels Bohr who proposed a novel model of the atom in 1913 which would lay the foundations for a quantum mechanical treatment ten years later. After discussing the limitations of Thomson's Plum Pudding model and Rutherford's Nuclear model, Bohr's q
From playlist Quantum Physics

Bohr Model (7 of 7) Electron Velocity Derivation and Calculation
This video shows how to calculate the electron velocities for each energy level of a hydrogen atom using the Bohr model. The velocities for the ground state and the first three excited states are calculated. Also shown is how each of the velocities can be converted into the energy in elect
From playlist Quantum Mechanics

Physics - Ch 66.5 Quantum Mechanics: The Hydrogen Atom (7 of 78) Other Radii Determined
Visit http://ilectureonline.com for more math and science lectures! In this video I will calculate and explain how the how the other radii were determined. From the previous video we calculated the inner most radius of the Bohr atom to be 53pm, so using the same principle of the de Brogli
From playlist PHYSICS 66.5 QUANTUM MECHANICS: THE HYDROGEN ATOM

Lec 4 | MIT 3.091 Introduction to Solid State Chemistry
Atomic Spectra of Hydrogen, Matter/Energy Interactions Involving Atomic Hydrogen View the complete course at: http://ocw.mit.edu/3-091F04 License: Creative Commons BY-NC-SA More information at http://ocw.mit.edu/terms More courses at http://ocw.mit.edu
From playlist MIT 3.091 Introduction to Solid State Chemistry, Fall 2004

Physics - Ch 66.5 Quantum Mechanics: The Hydrogen Atom (2 of 78) The Bohr Atom
Visit http://ilectureonline.com for more math and science lectures! In this video I will calculate the velocity v=?, frequency v=?, and period T=? of the Bohr Atom which is made up of a structure with a single positively charged small nucleus with an even smaller negatively charged electr
From playlist PHYSICS 66.5 QUANTUM MECHANICS: THE HYDROGEN ATOM

Bohr Model (6 of 7) Energy Level Derivation and Calculation
This video shows how to derive and calculate the energy levels of a hydrogen atom using the Bohr model. The energy of the ground state and the first three excited states are calculated. https://youtu.be/_kHJCX3hTaY Social Media for Step by Step Science: Teacher Pay Teachers Store: https:
From playlist Quantum Mechanics

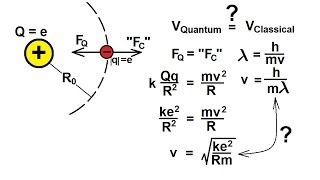
Physics - Modern Physics (16 of 26) The Bohr Atom: Velocity of the Electron
Visit http://ilectureonline.com for more math and science lectures! In this video I will show you how find the velocity of the electron of the Bohr Atom.
From playlist MODERN PHYSICS 2: ATOMIC AND NUCLEAR PHYSICS, PARTICLE PHYSICS