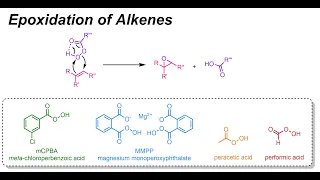
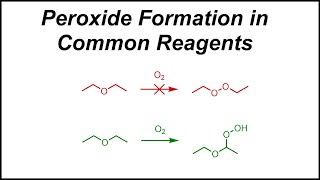
Peroxomonosulfate
The peroxomonosulfate ion, SO2−5, is a sulfur oxoanion. It is sometimes referred to as the persulfate ion, but this term also refers to the peroxydisulfate ion, S2O2−8. Its other IUPAC names are sulfuroperoxoate and trioxidoperoxidosulfate(2−). (Wikipedia).