
The Atom A3 The Bohr Model of the Hydrogen Atom
The Bohr model of the atom.
From playlist Physics - The Atom
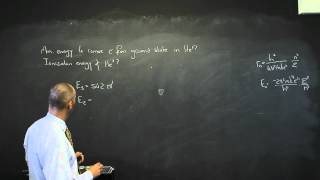
The Atom A5 The Bohr Model of the Hydrogen Atom
The Bohr model of the atom.
From playlist Physics - The Atom

The Atom A4 The Bohr Model of the Hydrogen Atom
The Bohr model of the atom.
From playlist Physics - The Atom

The Atom C3 The Pauli Exclusion Principle
The Pauli exclusion principle.
From playlist Physics - The Atom

The Atom B1 The Quantum Mechanical Picture of the Atom
The quantum mechanical model of the atom.
From playlist Physics - The Atom

The Atom C2 The Pauli Exclusion Principle
The Pauli exclusion principle.
From playlist Physics - The Atom

The Atom A1 Rutherford Scattering
The discovery of the nucleus of the atom.
From playlist Physics - The Atom

The Atom C1 The Pauli Exclusion Principle
The Pauli exclusion principle.
From playlist Physics - The Atom

The Atom B2 The Quantum Mechanical Picture of the Atom
The quantum mechanical model of the atom.
From playlist Physics - The Atom

Organic Chemistry 51B. Lecture 20. Radicals.
UCI Chem 51B Organic Chemistry (Winter 2013) Lec 20. Organic Chemistry -- Radicals View the complete course: http://ocw.uci.edu/courses/chem_51b_organic_chemistry.html Instructor: David Van Vranken, Ph.D. License: Creative Commons BY-NC-SA Terms of Use: http://ocw.uci.edu/info. More cour
From playlist Chemistry 51B: Organic Chemistry

Lec 20 | MIT 3.091 Introduction to Solid State Chemistry
Amorphous Solids, Glass Formation, Inorganic Glasses: Silicates View the complete course at: http://ocw.mit.edu/3-091F04 License: Creative Commons BY-NC-SA More information at http://ocw.mit.edu/terms More courses at http://ocw.mit.edu
From playlist MIT 3.091 Introduction to Solid State Chemistry, Fall 2004

Student Video: Hooke's Law in Cubic Solids
MIT RES.3-004 Visualizing Materials Science, Fall 2017 Speaker: anonymous MIT student View the complete course: https://ocw.mit.edu/RES-3-004F17 YouTube Playlist: https://www.youtube.com/playlist?list=PLUl4u3cNGP62EJXwSgoVRfh1tEiSc01bh Describes the behavior of springs using Hooke's Law.
From playlist MIT RES.3-004 Visualizing Materials Science, Fall 2017

Hooke's Law and Young's Modulus - A Level Physics
A description of Hooke's Law, the concepts of stress and strain, Young's Modulus (stress divided by strain) and energy stored in a stretched material
From playlist A Level Physics Revision

Lec 20 | MIT 3.091SC Introduction to Solid State Chemistry, Fall 2010
Lecture 20: Line, Interface & Bulk Defects Instructor: Donald Sadoway View the complete course: http://ocw.mit.edu/3-091SCF10 License: Creative Commons BY-NC-SA More information at http://ocw.mit.edu/terms More courses at http://ocw.mit.edu
From playlist MIT 3.091SC Introduction to Solid State Chemistry, Fall 2010

2. Force Laws, Lewis Structures and Resonance
Freshman Organic Chemistry (CHEM 125) Professor McBride begins by following Newton's admonition to search for the force law that describes chemical bonding. Neither direct (Hooke's Law) nor inverse (Coulomb, Gravity) dependence on distance will do - a composite like the Morse potential is
From playlist Freshman Organic Chemistry with J. Michael McBride

Nucleophiles and Electrophiles: Crash Course Organic Chemistry #12
Organic reactions are kind of like carefully choreographed fight scenes, and nucleophilic attack is a key move. This episode of Crash Course Organic Chemistry is all about nucleophiles and electrophiles, or what happens at those molecular hot spots we’ve been talking about. We’ll also lear
From playlist Organic Chemistry

20. Electronic and Vibrational Spectroscopy
Freshman Organic Chemistry II (CHEM 125B) Time-dependent quantum mechanics shows how mixing orbitals of different energy causes electrons to vibrate. Mixing 1s with 2p causes a vibration that can absorb or generate light, while mixing 1s with 2s causes "breathing" that does not interac
From playlist Freshman Organic Chemistry II with Michael McBride

Chem 203. Lecture 07: Fragmentation in EIMS Alkanes, Alkenes, Heteroatom Compounds, &Carbonyl Cmpds
Full Chem 203 Playlist: https://www.youtube.com/playlist?list=PLqOZ6FD_RQ7nUiPCa47zSrMWArKAdwfcD UCI Chem 203 Organic Spectroscopy (Fall 2020) Lecture 07: Fragmentation in EIMS Alkanes, Alkenes, Heteroatom Compounds, and Carbonyl Compounds Instructor: James S. Nowick, Ph.D. License: Creat
From playlist Chemistry 203, Organic Spectroscopy (2020)

Everything Matters: Germanium With Ron Hipschman
Appealingly shiny, nontoxic to mammals, and named for the home country of its discoverer, germanium is germane to any conversation about computer technology—it’s commonly used in the semiconductors that rule our computing lives. As part of the elemental series Everything Matters, join Expl
From playlist Tales from the Periodic Table

The Atom B3 The Quantum Mechanical Picture of the Atom
The quantum mechanical model of the atom.
From playlist Physics - The Atom