
Chemistry - Thermochemistry (1 of 37) Definition
Visit http://ilectureonline.com for more math and science lectures! In this video I will introduce of the definition of thermochemistry.
From playlist MOST POPULAR VIDEOS

What Is Thermosetting and Thermosoftening Polymers | Organic Chemistry | Chemistry | FuseSchool
Learn the basics about thermosetting and thermosoftening polymers, when learning about polymers as a part of organic chemistry. A polymer is a macromolecule made of many monomers, or repeating units. The properties of these polymers depend on a variety of factors – the monomer unit, the
From playlist CHEMISTRY

The Atom A3 The Bohr Model of the Hydrogen Atom
The Bohr model of the atom.
From playlist Physics - The Atom

Thermodynamic System | Open, Closed, Adiabatic, Isolated | Statistical Mechanics
In this video, we will define a thermodynamic system, in particular what kinds of thermodynamic systems there are and how they can interact with their surroundings. References: [1] Ansermet, Brechet, "Principles of Thermodynamics", Cambridge University Press (2019). Follow us on Insta
From playlist Thermodynamics, Statistical Mechanics

Thermodynamics: What do HEAT and WORK really mean? | Basics of Thermodynamics
"Work" and "heat" are commonly used words in everyday life. But they mean very specific things in the physics field of thermodynamics. Hey everyone, in this (rather short) video I wanted to discuss the exact definitions of the words "work" and "heat" in a thermodynamic context. These word
From playlist Thermodynamics by Parth G

The Atom A4 The Bohr Model of the Hydrogen Atom
The Bohr model of the atom.
From playlist Physics - The Atom

Physics - Thermodynamics: Temperature (1 of 3) What is the definition of Temperature?
Visit http://ilectureonline.com for more math and science lectures! In this video I will explain and give a definition of temperature as to how it relates to heat, kinetic energy, potential energy, and how it transfers.
From playlist MOST POPULAR VIDEOS
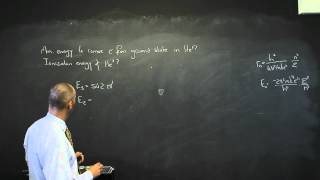
The Atom A5 The Bohr Model of the Hydrogen Atom
The Bohr model of the atom.
From playlist Physics - The Atom

Example: Application of Piecewise-Defined Functions
In this video, we work through an example of an application problem involving piecewise-defined functions.
From playlist College Algebra

Take-Home Problems #18 Solutions (corrected)
From playlist College Algebra

Take-Home Problems #18 Solutions
From playlist College Algebra

Ex: Estimate and Interpret the Slope of a Linear Cost Function
This video explains how to calculator and interpret the slope from the graph of a cost function.
From playlist Determining Linear Equations of Lines in Slope-intercept Form

T4D #120 - Lascar Winner and Chit Chat
Please support my content creation by using my Amazon Store: http://astore.amazon.com/m0711-20 ------------------------------ Click "Show more" ------------------------------------------- In this T4D I announce the winner of the Lascar Panel Pilot Ace SGD 43 - A giveaway. I show the Opgal
From playlist Tip or Thought for the Day!

Physics: Ch 24C -Heat Radiation: Test Your Knowledge (17 of 20) What is cost of Cooking with Gas?
Visit http://ilectureonline.com for more math and science lectures! http://www.ilectureonline.com/donate https://www.patreon.com/user?u=3236071 We will calculate the cost=? to boil 1 liter of water with the initial temperature of 20 degrees C assume $1 for each “therm”, where 1 therm=100
From playlist THE "WHAT IS" PLAYLIST

Thermal Camera Buyers Guide under $1500 - Pt 3
Support me through Patreon or by using my Amazon Store: Patreon: https://www.patreon.com/mjlorton Amazon Store: http://astore.amazon.com/m0711-20 Or anything on Amazon: http://www.amazon.com/?_encoding=UTF8&camp=212353&creative=380557&linkCode=sb1&tag=m0711-20 -----------------------------
From playlist Thermal Camera Buyers Guide

Thermal Camera Buyers Guide under $1500 - Pt 1
Support me through Patreon or by using my Amazon Store: Patreon: https://www.patreon.com/mjlorton Amazon Store: http://astore.amazon.com/m0711-20 Or anything on Amazon: http://www.amazon.com/?_encoding=UTF8&camp=212353&creative=380557&linkCode=sb1&tag=m0711-20 -----------------------------
From playlist Thermal Camera Buyers Guide

Thermal Camera Buyers Guide under $1500 - Pt 2
Support me through Patreon or by using my Amazon Store: Patreon: https://www.patreon.com/mjlorton Amazon Store: http://astore.amazon.com/m0711-20 Or anything on Amazon: http://www.amazon.com/?_encoding=UTF8&camp=212353&creative=380557&linkCode=sb1&tag=m0711-20 -----------------------------
From playlist Thermal Camera Buyers Guide

Lecture 14 - Solving Recurrences
This is Lecture 14 of the CSE547 (Discrete Mathematics) taught by Professor Steven Skiena [http://www.cs.sunysb.edu/~skiena/] at Stony Brook University in 1999. The lecture slides are available at: http://www.cs.sunysb.edu/~algorith/math-video/slides/Lecture%2014.pdf More information may
From playlist CSE547 - Discrete Mathematics - 1999 SBU

