
Valence Bond Theory, Hybrid Orbitals, and Molecular Orbital Theory
Attention! This video about molecular orbitals is much better: https://www.youtube.com/watch?v=I2k61JMk71M Alright, let's be real. Nobody understands molecular orbitals when they first take chemistry. You just pretend you do, and then in your next course you learn them a little better. An
From playlist General Chemistry

Valence Bond Theory & Hybrid Atomic Orbitals
This organic chemistry video tutorial provides a basic introduction into valence bond theory and hybrid atomic orbitals. It explains how to find the hybridization of carbon atom such as sp, sp2, and sp3. it also explains how to describe the orbitals that overlap to form a covalent bond.
From playlist New Organic Chemistry Playlist

Chemistry - Molecular Structure (23 of 45) Valance Bond Method
Visit http://ilectureonline.com for more math and science lectures! In this video I will explain the valence bonding method.
From playlist CHEMISTRY 14 MOLECULAR STRUCTURE

Concept of Valency - Introduction | Atoms And Molecules | Don't Memorise
If a bag of chocolates is kept open before us, we try grabbing as many chocolates as possible! Is that the same story with the Atoms? Do they also try grabbing as many Electrons as possible? Watch this video to get the answers! Valency is the number of atoms of a particular element that
From playlist Chemistry

Chemical Bonding | Covalent Bond | Ionic Bonding | Class 11 Chemistry
A chemical bond is a lasting attraction between atoms, ions or molecules that enables the formation of chemical compounds. The bond may result from the electrostatic force of attraction between oppositely charged ions as in ionic bonds or through the sharing of electrons as in covalent bon
From playlist Chemistry Topics

What Are Covalent Bonds | Properties of Matter | Chemistry | FuseSchool
What Are Covalent Bonds | Properties of Matter | Chemistry | FuseSchool Learn the basics about covalent bonds, when learning about properties of matter. When similar atoms react, like non-metals combining with other non-metals, they share electrons. This is covalent bonding. Non-metals
From playlist CHEMISTRY

Examples of s-p Mixing in Molecular Orbital Theory
Admittedly, my prior tutorial on MO theory was a little confusing, and had some errors. I wanted to make things right, so here's another one! This will clarify some of the basic concepts, and will also extend them to discuss a new concept, s-p mixing. Let's dive right in! Watch the whole
From playlist General Chemistry

Covalent Bond Energy and Length
We've already learned about different types of chemical bonds, including covalent bonds. But now that we know about enthalpy, and orbitals, and some other concepts, let's revisit the covalent bond and dig a little deeper. Watch the whole General Chemistry playlist: http://bit.ly/ProfDaveG
From playlist General Chemistry

A quick definition of valence. Chem Fairy: Louise McCartney Director: Michael Harrison Written and Produced by Kimberly Hatch Harrison ♦♦♦♦♦♦♦♦♦♦ Ways to support our channel: ► Join our Patreon : https://www.patreon.com/socratica ► Make a one-time PayPal donation: https://www.paypal.
From playlist Chemistry glossary

Lec 20 | MIT 5.112 Principles of Chemical Science, Fall 2005
Lewis and Bronsted Acid-Base Concepts View the complete course: http://ocw.mit.edu/5-112F05 License: Creative Commons BY-NC-SA More information at http://ocw.mit.edu/terms More courses at http://ocw.mit.edu
From playlist MIT 5.112 Principles of Chemical Science, Fall 2005

We've learned about VSEPR theory, and we know how to use it to predict molecular geometry for a variety of organic molecules. But in fact, there are situations where predictions made with VSEPR theory do not line up with experimental observation. Let's find out what these situations are, a
From playlist General Chemistry

The Chemical Bond: Covalent vs. Ionic and Polar vs. Nonpolar
Ionic Bond, Covalent Bond, James Bond, so many bonds! What dictates which kind of bond will form? Electronegativity values, of course. Let's go through each type and what they're all about. Watch the whole General Chemistry playlist: http://bit.ly/ProfDaveGenChem More AP Chemistry review
From playlist General Chemistry

Lec 13 | MIT 3.091 Introduction to Solid State Chemistry
Intrinsic and Extrinsic Semiconductors, Doping, Compound Semiconductors, Molten Semiconductors View the complete course at: http://ocw.mit.edu/3-091F04 License: Creative Commons BY-NC-SA More information at http://ocw.mit.edu/terms More courses at http://ocw.mit.edu
From playlist MIT 3.091 Introduction to Solid State Chemistry, Fall 2004

28. Modern Electronic Structure Theory: Basis Sets
MIT 5.61 Physical Chemistry, Fall 2017 Instructor: Professor Troy Van Voorhis View the complete course: https://ocw.mit.edu/5-61F17 YouTube Playlist: https://www.youtube.com/playlist?list=PLUl4u3cNGP62RsEHXe48Imi9-87FzQaJg Guest lecturer, Prof. Troy Van Voorhis, explores electronic struct
From playlist MIT 5.61 Physical Chemistry, Fall 2017

Doped Semiconductors Explained
The third in the series on semiconductors, this video explains how doping a semiconductor such as silicon, improves its conductive properties, using band theory model to explain. See www.physicshigh.com for all my videos and other resources. If you like this video, please press the LIKE a
From playlist Modern Physics

Natural semiconductors Explained
An explanation of how semiconductors work, in light of Band Theory. See www.physicshigh.com for all my videos and other resources. If you like this video, please press the LIKE and SHARE with your peers. And please add a COMMENT to let me know I have helped you. Follow me facebook: @phy
From playlist Modern Physics

MagLab Theory Winter School 2019: Rebecca Flint
Topic: Frustrated magnetism: models and magnetic order The National MagLab held it's seventh Theory Winter School in Tallahassee, FL from January 7th - 11th, 2019.
From playlist 2019 Theory Winter School

Deconfined quantum critical points: symmetries and dualities by Senthil Todadri
Date : Friday, July 7, 2017 Time : 3:00 PM Venue : Madhava Lecture Hall, ICTS Campus, Bangalore Abstract : It has been recognized for many years that at certain quantum phase transitions the standard Landau-Ginzburg-Wilson paradigm will break down. The most striking
From playlist Seminar Series

Concept of Valency | Atoms and Molecules | Don't Memorise
Do you know how Compounds are formed? From the Combination of Atoms right? But how and why do different Atoms combine? One of the crucial factors for this is Valency! Watch this video which explains how Valency plays an important role in the Formation of Compounds. To learn more about At
From playlist All About Atoms Class 09
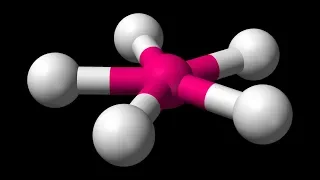
Visualizing Molecular Geometry With 3D Software
We've already learned about VSEPR theory, and how to use it to predict the shapes of various molecules. But we didn't cover all of the molecular geometries for certain hybridizations, and we didn't talk about bond angles, so let's do both of those things now, using some neat 3D software!
From playlist General Chemistry