
Chemistry - Liquids and Solids (13 of 59) Viscosity - Basics
Visit http://ilectureonline.com for more math and science lectures! In this video I will explain the basics of viscosity.
From playlist CHEMISTRY 16 LIQUIDS AND SOLIDS

Chemistry - Liquids and Solids (10 of 59) Surface Tension (Basics)
Visit http://ilectureonline.com for more math and science lectures! In this video I will explain the basics of surface tension.
From playlist CHEMISTRY 16 LIQUIDS AND SOLIDS

Physics demonstrations.Communicating vessels!
Communicating vessels is a stand alone, non enclosed system using a homogeneous fluid to help establish true level. The system will always settle and be at rest when the level in one vessel aligns with the level of the other in the system. There’s no limit to the number of vessels that can
From playlist PRESSURE

When two components are mixed, we need to do a binary phase diagram. The simplest of these is a isomorphous binary phase diagram. One example is the Ni-Cu system. In this system, we see a solidus line, liquidus line, and complete solutions in the liquid and solid phases.
From playlist Materials Sciences 101 - Introduction to Materials Science & Engineering 2020

Viscosity, Cohesive and Adhesive Forces, Surface Tension, and Capillary Action
Liquids have some very interesting properties, by virtue of the intermolecular forces they make, both between molecules of the liquid and those between the liquid and some other material they are in contact with. Let's learn about viscosity, cohesive and adhesive forces, surface tension, a
From playlist General Chemistry

Physics - Fluid Dynamics (1 of 25) Viscosity & Fluid Flow: Introduction
Visit http://ilectureonline.com for more math and science lectures! In this video I will introduce viscosity and fluid flow involving frictional forces between the molecules and the containing walls. Next video in this series can be seen at: https://youtu.be/3xukKynwA70
From playlist PHYSICS 34 FLUID DYNAMICS

23. Liquids: Brownian Motion and Forces in Liquids
MIT 2.57 Nano-to-Micro Transport Processes, Spring 2012 View the complete course: http://ocw.mit.edu/2-57S12 Instructor: Gang Chen License: Creative Commons BY-NC-SA More information at http://ocw.mit.edu/terms More courses at http://ocw.mit.edu
From playlist MIT 2.57 Nano-to-Micro Transport Processes, Spring 2012

DRY ICE vs ELECTRONIC COOLING!
We demonstrate why TECs or Peltier devices have a lower temperature limit. We also describe how low temperature liquid baths are set up in a lab, why they can be dangerous and even though we drop the "hot" side of a TEC to "only" -43C with a small amount of dry ice, we are approaching the
From playlist Cooling

Chemistry - Solutions (2 of 53) Six Types of Solutions
Visit http://ilectureonline.com for more math and science lectures! In this video I will explain the six types of solutions.
From playlist CHEMISTRY 19 SOLUTIONS

Luigi Preziosi: Multi-level mathematical models for cell migration in dense fibrous environments
Abstract: Cell-extracellular matrix interaction and the mechanical properties of cell nucleus have been demonstrated to play a fundamental role in cell movement across fibre networks and micro-channels and then in the spread of cancer metastases. The lectures will be aimed at presenting se
From playlist Mathematics in Science & Technology

Types of Colloids and Their Properties
Earlier we learned that as far as mixtures go, we can have homogeneous solutions, or totally heterogeneous mixtures, where components don't mix. But there are actually intermediary mixtures, where substances mix to some limited degree. Let's learn about colloids as well as suspensions! Wa
From playlist General Chemistry

Introduction to Solid State Physics, Lecture 14: Semiconductor Devices
Upper-level undergraduate course taught at the University of Pittsburgh in the Fall 2015 semester by Sergey Frolov. The course is based on Steven Simon's "Oxford Solid State Basics" textbook. Lectures recorded using Panopto, to see them in Panopto viewer follow this link: https://pitt.host
From playlist Introduction to Solid State Physics

Mod-01 Lec-16 Defect Structure & Mechanical Behaviour of Nanomaterials
Nanostructures and Nanomaterials: Characterization and Properties by Characterization and Properties by Dr. Kantesh Balani & Dr. Anandh Subramaniam,Department of Nanotechnology,IIT Kanpur.For more details on NPTEL visit http://nptel.ac.in.
From playlist IIT Kanpur: Nanostructures and Nanomaterials | CosmoLearning.org

24. Electrical Double Layer, Size Effects in Phase Change
MIT 2.57 Nano-to-Micro Transport Processes, Spring 2012 View the complete course: http://ocw.mit.edu/2-57S12 Instructor: Gang Chen License: Creative Commons BY-NC-SA More information at http://ocw.mit.edu/terms More courses at http://ocw.mit.edu
From playlist MIT 2.57 Nano-to-Micro Transport Processes, Spring 2012

Discussion Session by Richa Rikhy and Kaustubh Rane
DISCUSSION MEETING : THIRSTING FOR THEORETICAL BIOLOGY ORGANIZERS : Vijaykumar Krishnamurthy and Vidyanand Nanjundiah DATE : 03 June 2019 to 07 June 2019 VENUE : Ramanujan Lecture Hall, ICTS Bangalore Experimental biologists frequently point out that theoreticians trivialise th
From playlist Thirsting for Theoretical Biology 2019
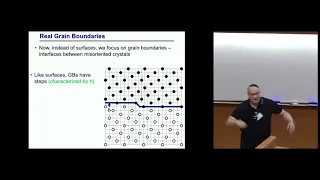
Macroscopic equations of motion for grain boundaries
David Srolovitz University of Pennsylvania, USA
From playlist 2018 Modeling and Simulation of Interface Dynamics in Fluids/Solids and Their Applications

Chemistry - Solutions (3 of 53) The Solution Process
Visit http://ilectureonline.com for more math and science lectures! In this video I will explain the solution process.
From playlist CHEMISTRY 19 SOLUTIONS

BUILD YOUR OWN REFRIGERATOR! - SOLID STATE TEC
We review various methods of refrigeration, focusing on thermoelectric coolers. We then explain the engineering and show the assembly of our solid state refrigerator. If you'd like to skip to the assembly - 37:01 Find us on Patreon - https://www.patreon.com/techingredients
From playlist Cooling

On the gradient-flow structure of multiphase mean curvature flow - Tim Laux
Analysis Seminar Topic: On the gradient-flow structure of multiphase mean curvature flow Speaker: Tim Laux Affiliation: University of California, Berkeley Date: December 9, 2019 For more video please visit http://video.ias.edu
From playlist Mathematics

What is a solution? | Solutions | Chemistry | Don't Memorise
What is a solution? You would say it is a mixture of two or more liquids. But is it so? Are solutions just mixtures of liquids? Watch this video to know the answers to these questions. In this video, we will learn: 0:00 What is a mixture? 0:49 properties of mixtures 2:08 Types of mixtur
From playlist Chemistry