
IB HL Chemistry Acids and bases Topic 18.1 Lewis acids and bases
IB HL Chemistry Acids and bases Topic 18.1 Lewis acids and bases A focused discussion on defining acids with the Lewis definition giving examples. Full set of resources for topic 8: http://www.mrwengibchemistry.com 0:10 Lewis acids and bases 2:44 Hydronium ion 4:08 Dative coordinate bon
From playlist Topic 8/18 Acids and bases

Finally, you'll understand all those weird pictures of molecules with the letters and the lines and the dots! Those are lewis dot structures. Let's learn how to draw them. It's easier than you think. Watch the whole General Chemistry playlist: http://bit.ly/ProfDaveGenChem More AP Chemis
From playlist General Chemistry

Mr. Andersen shows you how to draw Lewis Dot Diagrams for atoms and simple molecules. Intro Music Atribution Title: I4dsong_loop_main.wav Artist: CosmicD Link to sound: http://www.freesound.org/people/CosmicD/sounds/72556/ Creative Commons Atribution License
From playlist Chemistry

Imaginary numbers are any numbers that include the imaginary number i. A mix of imaginary and real numbers gives you what’s called a complex number. The primary reason we use imaginary numbers is to give us a way to find the root (radical) of a negative number. There’s no way to use real
From playlist Popular Questions

👉 Learn all about decimals. Decimals are numbers written with a decimal point. Digits can be written to the right or to the left of the decimal point. Digits are written to the left of the decimal point increase in value by multiples of 10 while digits written to the right decrease by mul
From playlist Decimals | Learn About

Lesson in senior chemistry on how to create Lewis dot diagrams
From playlist Chemistry

Chemistry - Chemical Bonding (2 of 35) Lewis Symbol for Ion2 and Ionic Compounds
Visit http://ilectureonline.com for more math and science lectures! In this video I will introduce the Lewis symbols for ions and ionic compounds.
From playlist CHEMISTRY 13 LEWIS STRUCTURES

Chemistry - Chemical Bonding (1 of 35) Lewis Symbol for Atoms
Visit http://ilectureonline.com for more math and science lectures! In this video I will introduce the Lewis symbols for atoms.
From playlist CHEMISTRY 13 LEWIS STRUCTURES

Lewis Structure: Chlorine Gas Cl2
Craig Beals shows how to draw the Lewis Structure for Chlorine Gas. This is a clip from the complete video: Covalent Bonding 2.1 - Drawing Lewis Structures
From playlist Chemistry Help

Lec 12 | MIT 5.111 Principles of Chemical Science, Fall 2005
Lewis Diagrams (Prof. Sylvia Ceyer) View the complete course: http://ocw.mit.edu/5-111F05 License: Creative Commons BY-NC-SA More information at http://ocw.mit.edu/terms More courses at http://ocw.mit.edu
From playlist MIT 5.111 Principles of Chemical Science, Fall 2005

Binary Tree 1. Constructing a tree (algorithm and pseudocode)
This is the first in a series of videos about binary trees. It is an explanation of the dynamic data structure known as the Binary Tree. It describes the way in which a binary tree is constructed, and how it can be represented numerically using a system of left and right pointers. This v
From playlist Data Structures

Lewis Structures, Introduction, Formal Charge, Molecular Geometry, Resonance, Polar or Nonpolar
This chemistry video tutorial explains how to draw lewis structures of molecules and the lewis dot diagram of polyatomic ions. It shows you how to calculate the formal charge, how to draw the resonance form of the lewis structure and how to determine the hybridization of the central atom.
From playlist New AP & General Chemistry Video Playlist

The True Story of the Tylenol Murders
Tamper-proof foil seals on medicine bottles have been commonplace for 40 years - but only came about after a series of random cyanide poisonings prompted their creation. Several people died as a result of this psychotic act that forever changed the face of over-the-counter medicine. Get to
From playlist American History

This chemistry video provides a basic introduction into how to draw lewis structures of common molecules such as Cl2, O2, OF2, CH4, NH3, H2O, C2H2, and N2H4. It contains a few examples and practice problems. My E-Book: https://amzn.to/3B9c08z Video Playlists: https://www.video-tutor.
From playlist New AP & General Chemistry Video Playlist

Chrome Office Hours: Performance
Join Paul Lewis and Paul Irish as they dive into some real world performance techniques that you can use to make your site screaming fast!
From playlist Development
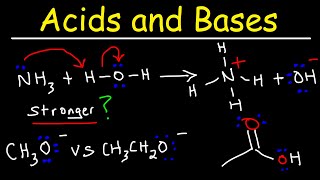
Acids and Bases - Basic Introduction - Organic Chemistry
This video provides a basic introduction into acids and bases with reference to organic chemistry. It explains how to write acid base reactions as well as how to determine the conjugate acid and the conjugate base. It covers the brownsted lowry definition of acids and bases as well as th
From playlist New Organic Chemistry Playlist

Lec 20 | MIT 5.112 Principles of Chemical Science, Fall 2005
Lewis and Bronsted Acid-Base Concepts View the complete course: http://ocw.mit.edu/5-112F05 License: Creative Commons BY-NC-SA More information at http://ocw.mit.edu/terms More courses at http://ocw.mit.edu
From playlist MIT 5.112 Principles of Chemical Science, Fall 2005

Exceptions To The Octet Rule - Lewis Dot Diagrams
This chemistry video tutorial discusses the exceptions to the octet rule while providing the lewis dot diagrams of the molecular compounds involved. BH3 has an incomplete octet - that is, it has less than 8 electrons. Molecules such as ICl5 and PCl5 have an expanded octet which means the
From playlist New AP & General Chemistry Video Playlist

Craig Beals shows how to draw the Lewis Structure for Ammonia. This is a clip from the complete video: Covalent Bonding 2.1 - Drawing Lewis Structures
From playlist Chemistry Help

ClO3- Lewis Structure - Chlorate Ion
This chemistry video tutorial explains how to draw the lewis structure of the Chlorate ion ClO3-. My Website: https://www.video-tutor.net Patreon: https://www.patreon.com/MathScienceTutor Amazon Store: https://www.amazon.com/shop/theorganicchemistrytutor Disclaimer: Some of the links
From playlist New AP & General Chemistry Video Playlist