
Law of Definite Proportions Chemistry Practice Problems - Chemical Fundamental Laws
This chemistry video tutorial discusses the law of definite proportions. It's a fundamental chemical law that states that the masses of each element in a given compound have constant composition. This tutorial contains plenty of examples and practice problems on how to find the number of
From playlist New AP & General Chemistry Video Playlist

Law of Definite Proportions - Law of multiple proportions
Watch more videos on http://www.brightstorm.com/science/chemistry SUBSCRIBE FOR All OUR VIDEOS! https://www.youtube.com/subscription_center?add_user=brightstorm2 VISIT BRIGHTSTORM.com FOR TONS OF VIDEO TUTORIALS AND OTHER FEATURES! http://www.brightstorm.com/ LET'S CONNECT! Facebook ► h
From playlist Chemistry

This video introduces the basis vocabulary and properties of proportions. Complete Video List at http://www.mathispower4u.com
From playlist Proportions

This video defines a proportion and explains how to solve a proportion. http://mathispower4u.wordpress.com/
From playlist Proportions

The Definition of The Definite Integral
This video defines a definite integral and provides examples of how to evaluate definite integral using area above and below the x-axis. Site: http://mathispower4u.com
From playlist Definite Integrals and The Fundamental Theorem of Calculus

Law of Multiple Proportions Practice Problems, Chemistry Examples, Fundamental Chemical Laws
This chemistry video tutorial explains the concept of the law of multiple proportions. It's another fundamental chemical law that states the ratio of the masses of the 2nd element for different compounds can be reduced to whole numbers. This video contains plenty of examples and practice
From playlist New AP & General Chemistry Video Playlist
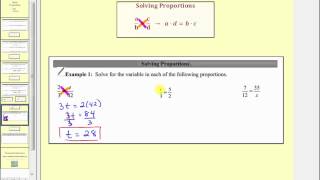
This video defines ratio, rate and proportion. Then several proportions are solved. Video content created by Jenifer Bohart, William Meacham, Judy Sutor, and Donna Guhse from SCC (CC-BY 4.0)
From playlist Proportions

Integration 4 The Definite Integral Part 3 Example 2
Working through another example of the definite integral.
From playlist Integration
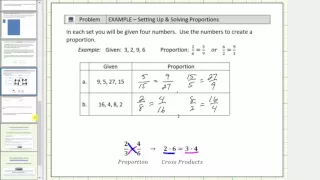
Proportions - Intro, Cross Product, and Applications
This video defines a proportion and then solves several problems using cross products.
From playlist Number Sense - Decimals, Percents, and Ratios

IGCSE Edexcel Physics lesson on particle nature of solids, liquids, and gases
From playlist Edexecel IGCSE Physics

[Lesson 20] QED Prerequisites: Field Potentials from the Principle of Least Action
This is a repost of Lesson 20 regarding the development of the electromagnetic field potential. The first post was low video quality and had some annoying errors! In this lesson we use insight from the Lorentz force law to guide us to finding an expression for the electromagnetic field po
From playlist QED- Prerequisite Topics

Mechanics 2 - Force, Mass & Acceleration
Arguably the most important equation in all of physics is F = ma, force equals mass times acceleration. Newton presented this law of Nature in 1687. Caltech's The Mechanical Universe episode Newton's Laws: https://youtu.be/tsJMfy2GH0A
From playlist Mechanics

Einstein's General Theory of Relativity | Lecture 1
Lecture 1 of Leonard Susskind's Modern Physics concentrating on General Relativity. Recorded September 22, 2008 at Stanford University. This Stanford Continuing Studies course is the fourth of a six-quarter sequence of classes exploring the essential theoretical foundations of modern ph
From playlist Lecture Collection | Modern Physics: Einstein's Theory

Law of Constant Proportions | Don't Memorise
Decomposition of any compound gives us the constituent elements. Did you know that the sum of masses of these elements is always exactly equal to the mass of the compound made out of them? That is what the Law of Constant Proportions tells us! Watch this video to get introduced to this Law
From playlist All About Atoms Class 09

Lecture 7 | Quantum Entanglements, Part 3 (Stanford)
Lecture 7 of Leonard Susskind's course concentrating on Quantum Entanglements (Part 3, Spring 2007). Recorded May 21, 2007 at Stanford University. This Stanford Continuing Studies course is the third of a three-quarter sequence of classes exploring the "quantum entanglements" in modern
From playlist Lecture Collection | Quantum Entanglements: Part 3 (Spring 2007)

Newton’s law of gravitation explained
Newton developed the Law of Gravitation as the force between two masses. This video covers the concept of Law of gravitation, gravitational field and uses and an example to help consolidate understanding. SLike what I do? Support by buying me a coffee - www.buymeacoffee.com/physicshigh S
From playlist Advanced Mechanics
Integration 4 The Definite Integral Part 3 Example 1
An example using the definite integral.
From playlist Integration

Deriving Hawking's most famous equation: What is the temperature of a black hole?
Black holes are perhaps the most enigmatic objects in the universe. Popularised in movies and science fiction, they evoke the magic and mystery of our universe and provide inspiration for those looking to make their mark in the world of academic physics. But what exactly is a black hole? A
From playlist Relativity