
In an ionic bond, two ions are held together by electrostatic force. These two atoms are trying to attain full valence shells of eight electrons. One atom has too many electrons and another has too few so one atom donates electrons and the other accepts them. The donor atom forms a cation,
From playlist Chemistry

Electric Field (2 of 3) Calculating the Magnitude and Direction of the Electric Field
Explains how to calculate the electric field of a charged particle and the acceleration of an electron in the electric field. You can see a listing of all my videos at my website, http://www.stepbystepscience.com An electric field is an area that surrounds an electric charge, and exerts f
From playlist Electricity and Magnetism

Electric Field (3 of 3) Calculating the Electric Field In Between Two Charges
Explains how to calculate the electric field between two charges and the acceleration of a charge in the electric field. You can see a listing of all my videos at my website, http://www.stepbystepscience.com An electric field is an area that surrounds an electric charge, and exerts force
From playlist Electricity and Magnetism

Electric Field (1 of 3) An Explanation
Explains how to determine the direction and magnitude of the electric field from charged particles. You can see a listing of all my videos at my website, http://www.stepbystepscience.com An electric field is an area that surrounds an electric charge, and exerts force on other charges in t
From playlist Electricity and Magnetism

Potential, Potential Difference, and Voltage
Gives a few of the basics of Electric Potential, Electric Potential Difference, and Voltage. For a complete index of these videos visit http://www.apphysicslectures.com
From playlist Phys 331 Videos - Youtube

Chemistry: What is an Ionic Bond? (Intramolecular Forces)
Practice Tests available at http://bit.ly/CHEMTESTS - 50 questions + Complete Answer Key Ionic bonds are one of the 3 main types of intramolecular forces, along with covalent bonds and metallic bonds. Ionic bonds are very strong, due to powerful electrostatic attraction between opposite i
From playlist Chemistry Lessons

Electric Potential In 30 Seconds!!
Can You Find The Electric Potential At The POSITION Of This Electron?!? #Electrical #Engineering #Physics #College #NicholasGKK #Shorts
From playlist Electrical Engineering

IB Chemistry Topic 4.1 Ionic bonding and structure
IB Chemistry Topic 4.1 Ionic bonding and structure How the ionic bond influences the physical and chemical properties of ionic compounds. How to draw ionic electron configurations and Lewis structures for ionic compounds. Describing ionic lattices. PPTs and quizzes are available on http:/
From playlist Topic 4/14 Chemical structure and bonding

Chemistry 107. Inorganic Chemistry. Lecture 13
UCI Chemistry: Inorganic Chemistry (Fall 2014) Lec 13. Inorganic Chemistry -- Ionic Structures View the complete course: http://ocw.uci.edu/courses/chem_107_inorganic_chemistry.html Instructor: Matthew D. Law License: Creative Commons CC-BY-SA Terms of Use: http://ocw.uci.edu/info More co
From playlist Chem 107: Week 5

Lattice Energy of Ionic Compounds, Basic Introduction, Charge vs Ionic Radius
This chemistry video tutorial provides a basic introduction into the lattice energy of ionic compounds. Lattice energy is the amount of energy released when 1 mole of gaseous ions combined to form a solid ionic compound. This video discusses the factors that affect the lattice energy of
From playlist New AP & General Chemistry Video Playlist

Unconventional superconductivity in half-filled ionic Hubbard model: Role of magnetic...by Arti Garg
PROGRAM FRUSTRATED METALS AND INSULATORS (HYBRID) ORGANIZERS Federico Becca (University of Trieste, Italy), Subhro Bhattacharjee (ICTS-TIFR, India), Yasir Iqbal (IIT Madras, India), Bella Lake (Helmholtz-Zentrum Berlin für Materialien und Energie, Germany), Yogesh Singh (IISER Mohali, In
From playlist FRUSTRATED METALS AND INSULATORS (HYBRID, 2022)

Atomic Bonding {Texas A&M: Intro to Materials}
Tutorial illustrating different types of bonding in solids. Ionic bonding, covalent bonding, metallic bonding, secondary bonding. Illustrated with examples. Video lecture for Introduction to Materials Science & Engineering (MSEN 201/MEEN 222), Texas A&M University, College Station, TX.
From playlist TAMU: Introduction to Materials Science & Engineering | CosmoLearning.org

Materialism Podcast Ep 31: Ionic conduction
Diffusion is when atoms move through a material in response to a concentration gradient. However, some ions can move through a material in response to an electric field and can do so very quickly! Even more remarkable, the motion of ions can be restricted to ions of a certain type such as
From playlist Materialism Podcast
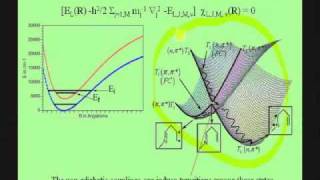
Lecture 01-Jack Simons Electronic Structure Theory- Born-Oppenheimer approximation
The Born-Oppenheimer approximation; non-adiabatic couplings; the electronic and vibration-rotation Schrodigner equations; atomic units; electronic cusps, electronic wave functions and energy surfaces; orbitals and spin-orbitals, Slater determinants; effects of antisymmetry; problems arisin
From playlist U of Utah: Jack Simons' Electronic Structure Theory course

How To Create A Flipboard Mobile App Clone | Ionic | Introduction
Don’t Forget To Hit Subscribe Button! This project series will teach you how to build a mobile app with Ionic. A clone of the Flipboard app. These project sessions will cover all the details (API, Tools, Languages, etc.) necessary to build a complete and operational Flipboard mobile ap
From playlist Create A Flipboard App Clone
Mod-06 Lec-16 Electrical Conduction in Ceramics ( Contd .)
Advanced ceramics for strategic applications by Prof. H.S. Maiti,Department of Metallurgy and Material Science,IIT Kharagpur.For more details on NPTEL visit http://nptel.ac.in
From playlist IIT Kharagpur: Advanced Ceramics for Strategic Applications | CosmoLearning.org Materials Science

Lec 12 | MIT 5.112 Principles of Chemical Science, Fall 2005
Ionic Bonds - Classical Model and Mechanism View the complete course: http://ocw.mit.edu/5-112F05 License: Creative Commons BY-NC-SA More information at http://ocw.mit.edu/terms More courses at http://ocw.mit.edu
From playlist MIT 5.112 Principles of Chemical Science, Fall 2005

Ionic crystal structures {Texas A&M: Intro to Materials}
Tutorial video illustrating "Ionic" crystal structures. How do we get these from ionic radius ratios? How do we figure out which sites are occupied? Video lecture for Introduction to Materials Science & Engineering (MSEN 201/MEEN 222), Texas A&M University, College Station, TX. http://e
From playlist TAMU: Introduction to Materials Science & Engineering | CosmoLearning.org

In the first mini-lecture, we define what a bond is. Are bonds static, or are they dynamic - moving about space more freely than a drawing of it might predict? We also define it by looking at the energetics when two atoms are brought closer and closer together. This is defined as Coulomb’s
From playlist Chemistry

Electric Potential: Visualizing Voltage with 3D animations
Shows how voltage can be visualized as electric potential energy. Includes topics such as why the voltage is the same everywhere inside a metal conductor.
From playlist Physics