Stability theory | Dynamical systems
Equilibrium point
In mathematics, specifically in differential equations, an equilibrium point is a constant solution to a differential equation. (Wikipedia).

Stability theory | Dynamical systems
In mathematics, specifically in differential equations, an equilibrium point is a constant solution to a differential equation. (Wikipedia).


Equilibrium occurs when the overall state of a system is constant. Equilibrium can be static (nothing in the system is changing), or dynamic (little parts of the system are changing, but overall the state isn't changing). In my video, I'll demonstrate systems in both types of equilibrium,
From playlist Physics

Stability Analysis, State Space - 3D visualization
Introduction to Stability and to State Space. Visualization of why real components of all eigenvalues must be negative for a system to be stable. My Patreon page is at https://www.patreon.com/EugeneK
From playlist Physics
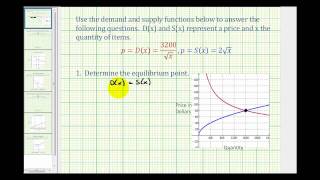
This video shows an example of how to determine the point of equilibrium given the supply and demand functions. Complete Video Library at www.mathispower4u.com
From playlist Business Applications of Integration

KS5 - Stationary & Turning Points
"Maxima and minima and stationary points."
From playlist Differentiation (AS/Beginner)

2D Equilibrium -- Balancing Games
How does everything even out? Learn what 2D Equilibrium is and how it effects the balance of life. License: Creative Commons BY-NC-SA More information at http://k12videos.mit.edu/terms-conditions
From playlist Measurement

http://mathispower4u.wordpress.com/
From playlist Applications of Definite Integration

From playlist l. Differential Calculus

Using the Second Derivative (2 of 5: Turning Point vs Stationary Point analogy)
More resources available at www.misterwootube.com
From playlist Applications of Differentiation

Senior Chemistry lesson on reaction kinetics and what the equilibrium constant represents and how to calculate.
From playlist Chemistry

Autonomous Equations, Equilibrium Solutions, and Stability
MY DIFFERENTIAL EQUATIONS PLAYLIST: ►https://www.youtube.com/playlist?list=PLHXZ9OQGMqxde-SlgmWlCmNHroIWtujBw Open Source (i.e free) ODE Textbook: ►http://web.uvic.ca/~tbazett/diffyqs Autonomous Differential Equations are ones of the form y'=f(y), that is only the dependent variable shows
From playlist Ordinary Differential Equations (ODEs)

IB HL Chemistry Lesson on equilibrium law and how can be applied to finding unknown concentrations.
From playlist IB Chemistry

Kousha Etessami: The complexity of computing a quasi perfect equilibrium for n player extensive form
We study the complexity of computing/approximating several classic refinements of Nash equilibrium for n-player extensive form games of perfect recall EFGPR, including perfect, quasi-perfect, and sequential equilibrium. We show that, for all of these refinements, approximating one such equ
From playlist HIM Lectures: Trimester Program "Combinatorial Optimization"

Lec 19 | MIT 5.111 Principles of Chemical Science, Fall 2005
Chemical Equilibrium (Prof. Catherine Drennan) View the complete course: http://ocw.mit.edu/5-111F05 License: Creative Commons BY-NC-SA More information at http://ocw.mit.edu/terms More courses at http://ocw.mit.edu
From playlist MIT 5.111 Principles of Chemical Science, Fall 2005

Mod-03 Lec-17 Different Aspects of Bertrand Model
Game Theory and Economics by Dr. Debarshi Das, Department of Humanities and Social Sciences, IIT Guwahati. For more details on NPTEL visit http://nptel.iitm.ac.in
From playlist IIT Guwahati: Game Theory and Economics | CosmoLearning.org Economics

18. Introduction to Chemical Equilibrium
MIT 5.111 Principles of Chemical Science, Fall 2014 View the complete course: https://ocw.mit.edu/5-111F14 Instructor: Catherine Drennan Reactions reach chemical equilibrium when the rate of the forward reaction equals the rate of the reverse reaction. In this lecture, we discuss the natu
From playlist MIT 5.111 Principles of Chemical Science, Fall 2014

Chemical Equilibrium Constant K - Ice Tables - Kp and Kc
This chemistry video tutorial provides a basic introduction into how to solve chemical equilibrium problems. It explains how to calculate the equilibrium constant k value given the equilibrium concentrations and equilibrium partial pressures of all reactants and products. If explains how
From playlist New AP & General Chemistry Video Playlist

Gömböc—The Shape That Shouldn't Exist
In this video I show you a Gömböc. This is a shape that has only two equilibrium points—one stable and one unstable, instead of the usual minimum of 4. It is self-righting no matter how you set it down. .STL file for the Gömböc: https://www.thingiverse.com/thing:523643 Checkout my expe
From playlist Amazing 3D Printed Objects

General Chemistry 1B. Lecture 15. Chemical Equilibrium, Part II
UCI Chem 1B General Chemistry (Winter 2013) Lec 15. General Chemistry -- Chemical Equilibrium Part 2 -- View the complete course: http://ocw.uci.edu/courses/chem_1b_general_chemistry.html Instructor: Donald R. Blake, Ph.D. License: Creative Commons BY-NC-SA Terms of Use: http://ocw.uci.ed
From playlist Chem 1B: Week 9