
Senior Chemistry lesson on calculation concentration of solutions
From playlist Chemistry

This video defines a ratio and provides several examples on how to write a ratio and shows how to simplify a ratio. http://mathispower4u.wordpress.com/
From playlist Ratios and Rates

Senior Chemistry lesson on reaction kinetics and what the equilibrium constant represents and how to calculate.
From playlist Chemistry

Chemistry - Solutions (6 of 53) Expressing Concentrations: Example 2
Visit http://ilectureonline.com for more math and science lectures! In this video I will find the mass of a prepare 3 liter of a 2% (W/V) saline solution.
From playlist CHEMISTRY 19 SOLUTIONS

System of 3 Equations with 3 Unknowns Application - Concentration Problem
This video provides an example of an application problem using a system of three equations with three unknowns. Site: http://mathispower4u.com
From playlist Systems of Equations with Three Unknowns
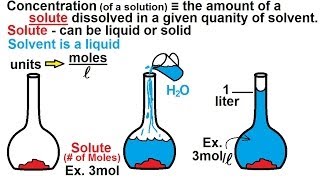
Chemistry - Concentration (Molarity) in Chemical Equations (25 of 38) : Definition
Visit http://ilectureonline.com for more math and science lectures! In this video I will show you how to calculate the definition of the concentration (molarity) of a solution.
From playlist CHEMISTRY 7 CONCENTRATION, REACTANTS, PRODUCTS

This video defines a proportion and explains how to solve a proportion. http://mathispower4u.wordpress.com/
From playlist Proportions

Chemistry - Acids & Bases Fundamentals (9 of 35) Calculating the pH: Ex 1
Visit http://ilectureonline.com for more math and science lectures! In this video I will calculate the pH given the concentration of H+.
From playlist CHEMISTRY 22 ACIDS AND BASES

Chemistry - Acids & Bases (30 of 45) Comparing Acid Strengths Using % Concentrations
Visit http://ilectureonline.com for more math and science lectures! In this video I will explain the % ionized of a strong, weak, weaker, and very weak acid.
From playlist CHEMISTRY 22 ACIDS AND BASES

Chemistry - Chemical Kinetics (11 of 30) Finding the Order - Using Initial Rates
Visit http://ilectureonline.com for more math and science lectures! In this video I will find the order using initial rates of multiple reactants.
From playlist CHEMISTRY 21 CHEMICAL KINETICS

IB HL Chemistry Lesson on equilibrium law and how can be applied to finding unknown concentrations.
From playlist IB Chemistry

We comparing different drink mixes using ratios
From playlist Middle School - Worked Examples

Ksp - Molar Solubility, Ice Tables, & Common Ion Effect
This chemistry video tutorial provides a basic introduction into Ksp - the solublity product constant. It explains how to calculate molar solubility from Ksp in mol/L and g/L and vice versa. It also explains how to solve common ion effect problems. Get The Full 1 Hour 26 Minute Video on
From playlist New AP & General Chemistry Video Playlist

General Chemistry 1C. Lecture 10. Buffered Solutions (Buffers) Pt. 1.
UCI Chem 1C General Chemistry (Spring 2013) Lec 10. General Chemistry -- Buffered Solutions (Buffers) -- Part 1 View the complete course: http://ocw.uci.edu/courses/chem_1c_general_chemistry.html Instructor: Ramesh D. Arasasingham, Ph.D. License: Creative Commons BY-NC-SA Terms of Use: ht
From playlist Chemistry 1C: General Chemistry

Chemistry - Chemical Kinetics (15 of 30) Finding Rate Law & Rate Constant, k
Visit http://ilectureonline.com for more math and science lectures! In this video I will determine the rate law and the rate constant, k.
From playlist CHEMISTRY 21 CHEMICAL KINETICS

Chemistry - Chemical Kinetics (5 of 30) Reaction Rate- Rate Law
Visit http://ilectureonline.com for more math and science lectures! In this video I will explain rate laws using the example N2O5,NO2, and O2.
From playlist CHEMISTRY 21 CHEMICAL KINETICS

General Chemistry 1C. Lecture 09. Acids and Bases. Pt. 6.
UCI Chem 1C General Chemistry (Spring 2013) Lec 09. General Chemistry -- Acids and Base -- Part 6 View the complete course: http://ocw.uci.edu/courses/chem_1c_general_chemistry.html Instructor: Ramesh D. Arasasingham, Ph.D. License: Creative Commons BY-NC-SA Terms of Use: http://ocw.uci.e
From playlist Chemistry 1C: General Chemistry

Rational Function Application - Concentration of a Mixture
This video explains how to write a rational function that gives the concentration of a mixture and than determine concentrations.
From playlist Determining Equations of Rational Functions

Chemistry - Solutions (8 of 53) Expressing Concentrations: Molarity: Example 1
Visit http://ilectureonline.com for more math and science lectures! In this video I will take a closer look at the effect of polarity on solubility.
From playlist CHEMISTRY 19 SOLUTIONS

ABR#16 Describing Acid Base Solutions
HSC Chemistry Acid/Base Reactions Describing Acid/Base Reactions Strength vs Concentration
From playlist Y12 Chem Mod 6 Acid/Base