
Senior Chemistry lesson on calculation concentration of solutions
From playlist Chemistry

W#16 Measures of concentration
Preliminary Chemistry Water Measures of concentration
From playlist Preliminary Chemistry - Water
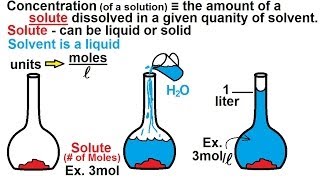
Chemistry - Concentration (Molarity) in Chemical Equations (25 of 38) : Definition
Visit http://ilectureonline.com for more math and science lectures! In this video I will show you how to calculate the definition of the concentration (molarity) of a solution.
From playlist CHEMISTRY 7 CONCENTRATION, REACTANTS, PRODUCTS

GCSE Science Revision Chemistry "Concentration of Solutions"
Find my revision workbooks here: https://www.freesciencelessons.co.uk/workbooks In this video, we look at how to calculate the concentration of a solution and then the effect of changing the mass of solute and the volume of solution on the concentration.
From playlist 9-1 GCSE Chemistry Paper 1 Quantitative Chemistry

GCSE Science Revision Chemistry "Using Concentration of Solutions 1" (Triple)
Find my revision workbooks here: https://www.freesciencelessons.co.uk/workbooks In this video, we learn how to calculate the concentration of a solution in moles per decimetre cubed. We learn how to use this to work out the mass of a solute dissolved in a solvent.
From playlist 9-1 GCSE Chemistry Paper 1 Quantitative Chemistry

From playlist h. Three-Dimensional Measurement

Practice Problem: Calculating Equilibrium Concentrations
We know how to write equilibrium constant expressions for any equilibrium, and we know that they relate the equilibrium constant to the equilibrium concentrations for all of the substances in the system. Let's see if we can calculate a missing equilibrium concentration when other data is g
From playlist General Chemistry Practice Problems

Chemistry - Acids & Bases Fundamentals (9 of 35) Calculating the pH: Ex 1
Visit http://ilectureonline.com for more math and science lectures! In this video I will calculate the pH given the concentration of H+.
From playlist CHEMISTRY 22 ACIDS AND BASES

Chemistry - Solutions (4 of 53) Expressing Concentrations
Visit http://ilectureonline.com for more math and science lectures! In this video I will explain how concentrations are expressed in solutions.
From playlist CHEMISTRY 19 SOLUTIONS

MIT 5.111 Principles of Chemical Science, Fall 2014 View the complete course: https://ocw.mit.edu/5-111F14 Instructor: Catherine Drennan Whether a reaction will go forward spontaneously depends on the thermodynamics. How fast a reaction goes depends on the kinetics. Decomposition of a mol
From playlist MIT 5.111 Principles of Chemical Science, Fall 2014

General Chemistry 1C. Lecture 23. Chemical Kinetics Pt. 2.
UCI Chem 1C General Chemistry (Spring 2013) Lec 23. General Chemistry -- Chemical Kinetics -- Part 2 View the complete course: http://ocw.uci.edu/courses/chem_1c_general_chemistry.html Instructor: Ramesh D. Arasasingham, Ph.D. License: Creative Commons BY-NC-SA Terms of Use: http://ocw.uc
From playlist Chemistry 1C: General Chemistry

Introduction to Chemical Engineering | Lecture 18
Professor Channing Robertson of the Stanford University Chemical Engineering Department discusses the functioning of the kidney, focusing upon the single nephron glomeruli filtration rate, SNGFR. Introduction to Chemical Engineering (E20) is an introductory course offered by the Stanfor
From playlist Lecture Collection | Introduction to Chemical Engineering

Limitations to Measurements in Cellular Systems by Vaibhav Wasnik
DISCUSSION MEETING 8TH INDIAN STATISTICAL PHYSICS COMMUNITY MEETING ORGANIZERS: Ranjini Bandyopadhyay (RRI, India), Abhishek Dhar (ICTS-TIFR, India), Kavita Jain (JNCASR, India), Rahul Pandit (IISc, India), Samriddhi Sankar Ray (ICTS-TIFR, India), Sanjib Sabhapandit (RRI, India) and Prer
From playlist 8th Indian Statistical Physics Community Meeting-ispcm 2023

Lec 31 | MIT 5.111 Principles of Chemical Science, Fall 2005
Kinetics (Prof. Catherine Drennan) View the complete course: http://ocw.mit.edu/5-111F05 License: Creative Commons BY-NC-SA More information at http://ocw.mit.edu/terms More courses at http://ocw.mit.edu
From playlist MIT 5.111 Principles of Chemical Science, Fall 2005

Radek Adamczak: Functional inequalities and concentration of measure II
Concentration inequalities are one of the basic tools of probability and asymptotic geo- metric analysis, underlying the proofs of limit theorems and existential results in high dimensions. Original arguments leading to concentration estimates were based on isoperimetric inequalities, whic
From playlist Winter School on the Interplay between High-Dimensional Geometry and Probability

R2. Pre-Steady State and Steady-State Kinetic Methods Applied to Translation
MIT 5.08J Biological Chemistry II, Spring 2016 View the complete course: https://ocw.mit.edu/5-08JS16 Instructor: JoAnne Stubbe Methods to monitor the role of EF-Tu in translation is the focus of this recitation. The use of radioactivity will be introduced. License: Creative Commons BY-N
From playlist MIT 5.08J Biological Chemistry II, Spring 2016

Frontiers of Biomedical Engineering (BENG 100) Professor Saltzman continues his description of nephron anatomy, and the specific role of each part of the nephron in establishing concentration gradients to help in secretion and reabsorption of water, ions, nutrients and wastes. A number
From playlist Frontiers of Biomedical Engineering with W. Mark Saltzman

Mod-11 Lec-26 Ceramic Gas Sensor
Advanced ceramics for strategic applications by Prof. H.S. Maiti,Department of Metallurgy and Material Science,IIT Kharagpur.For more details on NPTEL visit http://nptel.ac.in
From playlist IIT Kharagpur: Advanced Ceramics for Strategic Applications | CosmoLearning.org Materials Science

Chemistry - Solutions (8 of 53) Expressing Concentrations: Molarity: Example 1
Visit http://ilectureonline.com for more math and science lectures! In this video I will take a closer look at the effect of polarity on solubility.
From playlist CHEMISTRY 19 SOLUTIONS

Thermodynamics and Chemical Dynamics 131C. Lecture 19. Observational Chemical Kinetics
UCI Chem 131C Thermodynamics and Chemical Dynamics (Spring 2012) Lec 19. Thermodynamics and Chemical Dynamics -- Observational Chemical Kinetics -- View the complete course: http://ocw.uci.edu/courses/chem_131c_thermodynamics_and_chemical_dynamics.html Instructor: Reginald Penner, Ph.D.
From playlist Chemistry 131C: Thermodynamics and Chemical Dynamics