
Carbenes Part 1: Properties and Formation
We've learned about a variety of different unstable carbon species, like carbocations, carbanions, and carbon radicals. But there is one more to know, carbenes. These are carbon atoms with two bonds and two nonbonding electrons. They have some interesting properties and applications so let
From playlist Organic Chemistry

Carbenes Part 2: Cyclopropanation, C-H Insertion, and the Bamford-Stevens Reaction
In the previous tutorial we learned about carbenes. So now that we understand their properties, we are ready to learn about what we can do with them. Three major uses of carbenes involve cyclopropanation, C-H insertion, and the Bamford-Stevens reaction. Let's see what these are now! Watch
From playlist Organic Chemistry

We know how to do acid-catalyzed hydration, which is a Markovnikov hydration, but there is another way to do this, one that does not result in carbocation rearrangement. Let's look at the mechanism and application of oxymercuration-demercuration. Watch the whole Organic Chemistry playlist
From playlist Organic Chemistry

This organic chemistry video tutorial provides a basic introduction into functional groups. It covers alkanes, alkenes, alkynes, aromatic rings, alcohols, ethers, esters, carboxylic acids, ketones, aldehydes, acid chlorides, acid anhydrides, amines, amides, nitriles, thiols, thioethers, t
From playlist New Organic Chemistry Playlist

Retrosynthesis 7 - Organic Chemistry
An introductory level video for retrosynthesis using 1,5-diX disconnections in organic chemistry. These disconnections often involved conjugate addition (Michael addition) using enamines or 1,3-dicarbonyl reagents. #chemistry #organicchemistry #orgo #retrosynthesis #synthesis #stem #steme
From playlist Retrosynthesis

Carbocation Rearrangement - Hydride and Methanide Shifts
Hydride and methanide shifts? What are those? Well, I'll tell you. Surely by now you've noticed that's what I'm doing here, no? Carbocation rearrangement is no exception. Watch the whole Organic Chemistry playlist: http://bit.ly/ProfDaveOrgChem General Chemistry Tutorials: http://bit.ly/
From playlist Organic Chemistry

Retrosynthesis 4 - Organic Chemistry
A few disconnection strategies for the retrosynthesis of this conjugated ketone. This video shows how you can split up a 1,4-diX functional group relationship in different ways that use umpolung chemistry. #chemistry #organicchemistry #orgo #ochem #organic #retrosynthesis There is an eno
From playlist Retrosynthesis

Synthesis Workshop: Synthesis of Tomatidine with Chad Normandin (Episode 105)
In this Research Spotlight episode, Chad Normandin (Barriault group, University of Ottawa) joins us to share his work on the synthesis of tomatidine. Key references: Eur. J. Org. Chem. 2020, 2693-2698. https://doi.org/10.1002/ejoc.202000051 Molecules 2021, 26, 6008. https://doi.org/10.339
From playlist Research Spotlights

Synthesis Workshop: Aziridine Synthesis via Electrogenerated Dications with Dylan Holst (Episode 66)
In this Research Spotlight episode, we're joined by Dylan Holst, who shares with us his work on aziridine synthesis via electrogenerated dications. Key reference: Nature 2021, 596, 74-79. https://doi.org/10.1038/s41586-021-03717-7 Also see: Nat. Chem. 2021. https://doi.org/10.1038/s41557-
From playlist Research Spotlights

Carbohydrates Part 2: Polysaccharides
Now that we know about simple sugars, we need to know about how these polymerize to form long polysaccharide chains, like cellulose, starch, and glycogen. Check it out! Watch the whole Biochemistry playlist: http://bit.ly/ProfDaveBiochem General Chemistry Tutorials: http://bit.ly/ProfDav
From playlist Biochemistry
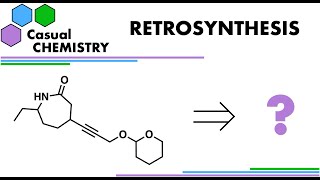
Retrosynthesis 10 - Organic Chemistry
Retrosynthetic analysis of a substituted caprolactam, highlighting the Beckmann rearrangement, Michael addition, and protecting group chemistry using the THP group. #chemistry #organicchemistry #orgo #synthesis #ochem #retrosynthesis #stem #education #science The 7-membered ring in this
From playlist Retrosynthesis

Carbocation Stability - Hyperconjugation, Inductive Effect & Resonance Structures
This organic chemistry video tutorial provides a basic introduction into carbocation stability. It discusses hyperconjugation and the inductive effect of electron donating groups and electron withdrawing groups. It also discusses carbocation stability using resonance structures. SN1 SN2
From playlist New Organic Chemistry Playlist

Total Synthesis Of Dimeric Securinega Alkaloids Flueggenines D And I Part One
A lecture on the Total Synthesis Of Dimeric Securinega Alkaloids Flueggenines D And I as reported by Sangbin Jeon, Jinwoo Lee, Sangbin Park and Sunkyu Han*. References: Chem. Sci., 2020,11, 10934-10938 (This work) Chem. Commun., 2014,50, 9284-9287 (Synthesis of derivatized proline) J. Am.
From playlist Total Synthesis

Watch more videos on http://www.brightstorm.com/science/biology SUBSCRIBE FOR All OUR VIDEOS! https://www.youtube.com/subscription_center?add_user=brightstorm2 VISIT BRIGHTSTORM.com FOR TONS OF VIDEO TUTORIALS AND OTHER FEATURES! http://www.brightstorm.com/ LET'S CONNECT! Facebook ► htt
From playlist Biology
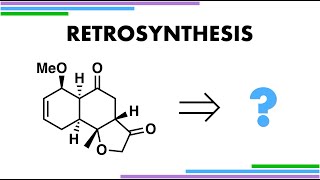
Retrosynthesis with Hypervalent Iodine
Disconnecting this tricyclic ring molecule using hypervalent iodine reagents. Also showcasing Umpolung chemistry and the Diels-Alder reaction. Reference: Desymmetrisation of phenols by an asymmetric Stetter reaction (Rovis) J. Am. Chem. Soc. 2006, 128, 8, 2552–2553 https://doi.org/10.1021
From playlist Retrosynthesis

Retrosynthesis 3 - Organic Chemistry
A retrosynthesis of the diketone known as dimedone. The functionality suits disconnections that use enolate and conjugate addition routes. #organicchemistry #chemistry #orgo #ochem #retrosynthesis Retrosynthetic analysis of dimedone shows that there is both a 1,3-difunctional and 1,5-dif
From playlist Retrosynthesis

This video is an overview on the four main organic compounds: proteins, lipids, nucleic acids, and carbohydrates. Questions? Feel free to post them in the comments and I'll do my best to answer!
From playlist Biology

A Pyridine Dearomatization Approach to the Matrine-type Lupin Alkaloids with Jeff Kerkovius
In this Research Spotlight episode, Jeff Kerkovius (Reisman group, Caltech) takes us through his work on a pyridine dearomatization approach to the synthesis of matrine-type lupin alkaloids. Key reference: J. Am. Chem. Soc. 2022, 144, 15938-15943. https://doi.org/10.1021/jacs.2c06584 Add
From playlist Research Spotlights

Allylic/Benzylic Bromination With N-Bromo Succinimide (NBS)
We've learned about radical bromination for alkanes, but there is another method of radical bromination that can be applied to allylic and benzylic systems. This utilizes the reagent NBS, and it can be a useful synthetic technique. Let's check out the transformation this induces as well as
From playlist Organic Chemistry

Synthesis Workshop: Selective Dearomatization of Phenols with Prof. Sarah Wengryniuk (Episode 71)
In this Research Spotlight episode, Prof. Sarah Wengryniuk joins us to talk about selective dearomatization of phenols using hypervalent iodine reagents. This episode has been made possible through a collaboration with Thieme Chemistry (Twitter: @thiemechemistry) and is made in recognition
From playlist Research Spotlights